8-K
False0001520262Alkermes plc.00015202622025-09-082025-09-08
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(d) OF THE
SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of earliest event reported): September 8, 2025
ALKERMES PUBLIC LIMITED COMPANY
(Exact name of registrant as specified in its charter)
|
|
|
|
|
|
|
Ireland |
|
001-35299 |
|
98-1007018 |
(State or other jurisdiction |
|
(Commission |
|
(IRS Employer |
of incorporation) |
|
File Number) |
|
Identification No.) |
|
|
|
|
|
|
Connaught House, 1 Burlington Road |
Dublin 4, Ireland D04 C5Y6 |
(Address of principal executive offices) |
Registrant's telephone number, including area code: + 353-1-772-8000
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
|
|
|
☐ |
|
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
|
|
|
☐ |
|
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
|
|
|
☐ |
|
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
|
|
|
☐ |
|
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class |
|
Trading Symbol(s) |
|
Name of each exchange on which registered |
Ordinary shares, $0.01 par value |
|
ALKS |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
|
|
|
|
|
Emerging growth company ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01 Regulation FD Disclosure.
On September 8, 2025, in connection with its participation at the World Sleep Congress taking place September 5-10, 2025, Alkermes plc (the “Company”) issued a press release announcing detailed safety and efficacy results from Vibrance-1, the Company’s phase 2 study evaluating the safety and efficacy of alixorexton (formerly referred to as ALKS 2680) compared to placebo in patients with narcolepsy type 1. Copies of the Company’s press release and related investor presentation and scientific presentations are furnished herewith as Exhibit 99.1, Exhibit 99.2 and Exhibit 99.3, respectively, and are incorporated herein by reference.
The information in this Item 7.01, and in Exhibit 99.1, Exhibit 99.2 and Exhibit 99.3 furnished herewith, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such a filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
EXHIBIT INDEX
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
ALKERMES PLC |
|
|
Date: September 8, 2025 |
By: |
|
/s/ David J. Gaffin |
|
|
|
David J. Gaffin |
|
|
|
Secretary |
EX-99.1
Exhibit 99.1
|
|
|
|
Alkermes Contacts: |
|
|
For Investors: |
Sandy Coombs, +1 781 609 6377 |
|
For Media: |
Gretchen Murphy,+1 781 609 6419 |
Alkermes Presents Detailed Positive Results from Vibrance-1 Phase 2 Study of Alixorexton in Patients with Narcolepsy Type 1 at World Sleep Congress 2025
– First Orexin 2 Receptor Agonist to Demonstrate Clinically Meaningful and Statistically Significant Impact on Wakefulness, Cognition and Fatigue with Once-Daily Dosing Across a Range of Doses –
– Alixorexton Was Generally Well Tolerated at All Doses Tested –
– Company to Host Investor Webcast on Monday, Sept. 8 at 8:00 a.m. ET –
DUBLIN, Sept. 8, 2025 -- Alkermes plc (Nasdaq: ALKS) today announced detailed positive results from the Vibrance-1 phase 2 study evaluating alixorexton in patients with narcolepsy type 1 (NT1). Alixorexton, formerly ALKS 2680, is a novel, investigational, oral, selective orexin 2 receptor (OX2R) agonist in phase 2 development as a once-daily treatment for NT1, narcolepsy type 2 (NT2) and idiopathic hypersomnia (IH). The randomized, placebo-controlled, six-week, double-blind phase 2 study conducted in 92 patients with NT1 demonstrated clinically meaningful and statistically significant improvements in wakefulness, cognition and fatigue that were sustained over the six-week treatment period. Alixorexton was generally well tolerated at all doses tested (4 mg, 6 mg and 8 mg).
“The detailed Vibrance-1 dataset presented at World Sleep highlights the robust efficacy of once-daily alixorexton in improving wakefulness and reducing excessive daytime sleepiness in patients with narcolepsy type 1, along with its generally well tolerated safety profile. The improvements in patient-reported outcomes, especially those related to fatigue and cognitive function, suggest that alixorexton may offer meaningful relief across a spectrum of symptoms that impact patients,” said Giuseppe Plazzi, M.D., Ph.D., Neurologist, Director of the Narcolepsy Center at the IRCCS of the Neurological Sciences of Bologna and Professor of Childhood Neuropsychiatry at the University of Modena and Reggio Emilia. “These data underscore alixorexton’s potential to be an important new treatment option for narcolepsy type 1 and to reduce the broader disease burden of this complex neurological disorder.”
The data were presented in three oral presentations at World Sleep Congress, taking place Sept. 5-10, 2025 in Singapore. Prespecified analyses are included in the table below. Key highlights include:
•Once-daily alixorexton met the primary endpoint across all doses tested, demonstrating statistically significant, clinically meaningful and dose-dependent improvements from baseline compared to placebo in mean sleep latency (MSL) on the Maintenance of Wakefulness Test (MWT) at week six. Patients had a mean sleep latency of approximately 3 minutes at baseline. All alixorexton dose groups achieved normative wakefulness on the MWT (mean sleep latency ≥20 minutes), with observed mean sleep latency of approximately 24 minutes, 26 minutes and 28 minutes for the 4, 6 and 8 mg doses, respectively.
•Alixorexton demonstrated robust and clinically meaningful improvements on the key secondary endpoint evaluating change from baseline versus placebo on the Epworth Sleepiness Scale (ESS) at week six.1 Patients had a mean ESS score of 18.5 at baseline. Improvements in ESS were sustained in the normal range (a score of ≤10) for all doses tested across all timepoints during the six-week double-blind treatment period and the subsequent open-label extension period through week 13.2
•On the key secondary endpoint evaluating mean weekly cataplexy rates, alixorexton demonstrated numerical and clinically meaningful improvements across all doses compared to placebo at weeks five and six3 and, on the pre-specified analysis, achieved statistical significance at the 6 mg dose. More than 40% of patients at the 6 mg and 8 mg doses achieved 100% reduction in cataplexy during week six of the study.
•Vibrance-1 also included a range of exploratory patient-reported outcome measures. Alixorexton drove statistically significant and clinically meaningful improvements from baseline compared to placebo in disease severity, fatigue and cognitive impairment. At week six, most patients receiving alixorexton reported mild narcolepsy severity.4Across all timepoints and all alixorexton dose groups, mean cognitive impairment scores fell within the lowest severity category of “none or minimal” impairment and mean fatigue scores fell into the “normal” range—effectively achieving normalization across both cognition and fatigue.5,6
|
|
|
|
|
Primary and Key Secondary Endpoints Reported p-values adjusted for multiplicity |
Change from Baseline at
Week 6 vs. Placebo |
Placebo |
4 mg |
6 mg |
8 mg |
LSM* |
LSM vs. placebo |
MSL on MWT (minutes) |
-0.6 |
22.2 p=0.01 |
24.1 p<0.0001 |
26.0 p<0.0001 |
ESS |
-3.1 |
-6.4 p=0.01 |
-8.7 p<0.0001 |
-8.3 p<0.0001 |
Weekly Cataplexy Rate
(Rate ratio vs. placebo at weeks 5 and 6)3 |
-- |
0.49 p=0.169 |
0.31 p=0.01 |
0.64 p=0.452 |
|
|
|
|
|
Patient-reported Outcomes Exploratory endpoints; Reported p-values are nominal |
Change from Baseline at
Week 6 vs. Placebo |
Placebo |
4 mg |
6 mg |
8 mg |
LSM |
LSM vs. placebo |
Narcolepsy Severity Scale for Clinical Trials (NSS-CT)4 |
-7.1 |
-9.1 p=0.0008 |
-12.4 p<0.0001 |
-11.0 p<0.0001 |
PROMIS-Fatigue5 |
-3.3 |
-8.7 p=0.0018 |
-12.4 p<0.0001 |
-12.9 p<0.0001 |
British Columbia Cognitive Complaints Inventory (BC-CCI)6 |
-1.2 |
-3.5 p<0.0001 |
-3.7 p<0.0001 |
-4.8 p<0.0001 |
*Least-squares mean difference
•Alixorexton was generally well tolerated across all doses tested throughout the six-week, randomized, double-blind treatment period. No serious treatment-emergent adverse events (TEAEs) were reported. There were no clinically meaningful changes in hepatic and renal parameters, vital signs, ECGs or ophthalmic exams in the alixorexton-treated group.
•Most TEAEs were mild to moderate in severity. The most common TEAEs7 were pollakiuria, insomnia, salivary hypersecretion, urinary urgency and blurred vision. Events of insomnia largely occurred and resolved within the first week of dosing. Events of blurred vision were mostly mild and intermittent and largely occurred and resolved within the first three days of treatment.
•More than 95% of patients who participated in the six-week double-blind portion of the trial entered into the seven-week open-label extension.
“As we seek to unlock a new era of innovation in neuroscience, the compelling results from Vibrance-1 underscore the strength of Alkermes’ orexin program. These data represent a significant new contribution to the evidence base supporting the utility of orexin 2 receptor agonists in central disorders of hypersomnolence and support exploration of the broader therapeutic potential of the class across a range of psychiatric and neurological conditions,” said Richard Pops, Chief Executive Officer of Alkermes. “We believe orexin-targeted therapeutics represent a significant opportunity for growth. We look forward to advancing alixorexton into phase 3 as soon as possible, and ALKS 4510 and ALKS 7290 into first-in-human studies this year, with the goal of delivering novel and differentiated new treatments across a broad range of disorders.”
Based on these results, Alkermes plans to initiate a global phase 3 program for alixorexton in the first quarter of 2026. Vibrance-2, a phase 2 study evaluating the safety and efficacy of alixorexton in adults with NT2 (NCT06555783), recently completed enrollment. Vibrance-3, a phase 2 study evaluating the safety and efficacy of alixorexton in adults with IH (NCT06843590), is currently enrolling.
Conference Call and Webcast
Alkermes will host a webcast presentation and conference call with accompanying slides for analysts and investors on Monday, Sept. 8, 2025, at 8:00 a.m. ET (8:00 p.m. SGT) to discuss these data. The webcast player may be accessed on the Investors section of Alkermes’ website at www.alkermes.com. To participate in the question-and-answer session, please also dial in to the conference call, which may be accessed by dialing +1 877-407-2988 for U.S. callers and +1 201-389-0923 for international callers. A replay of the webcast will be archived on the company’s website for 30 days following the presentation.
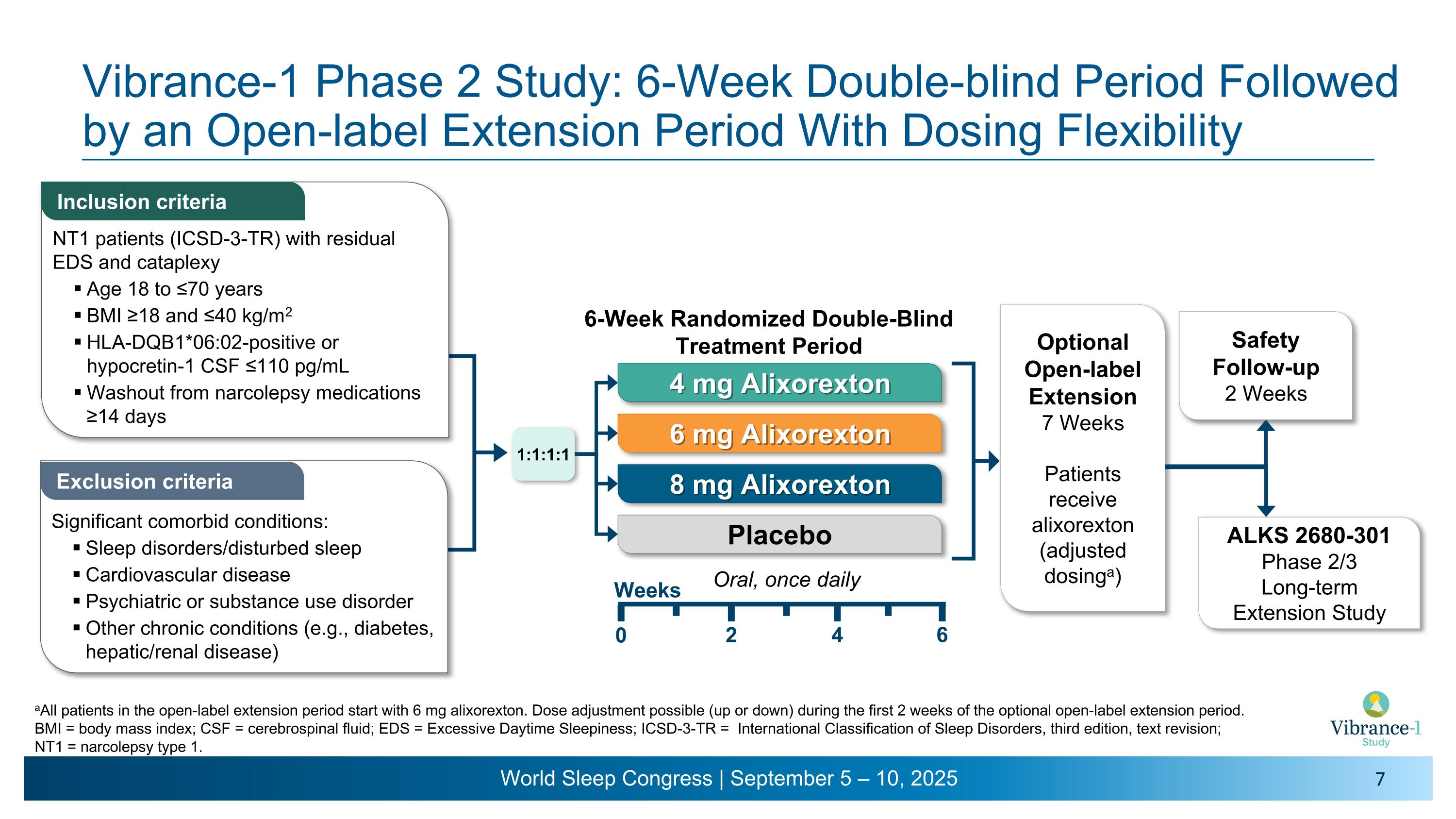
About the Vibrance-1 Phase 2 Study (NCT06358950)
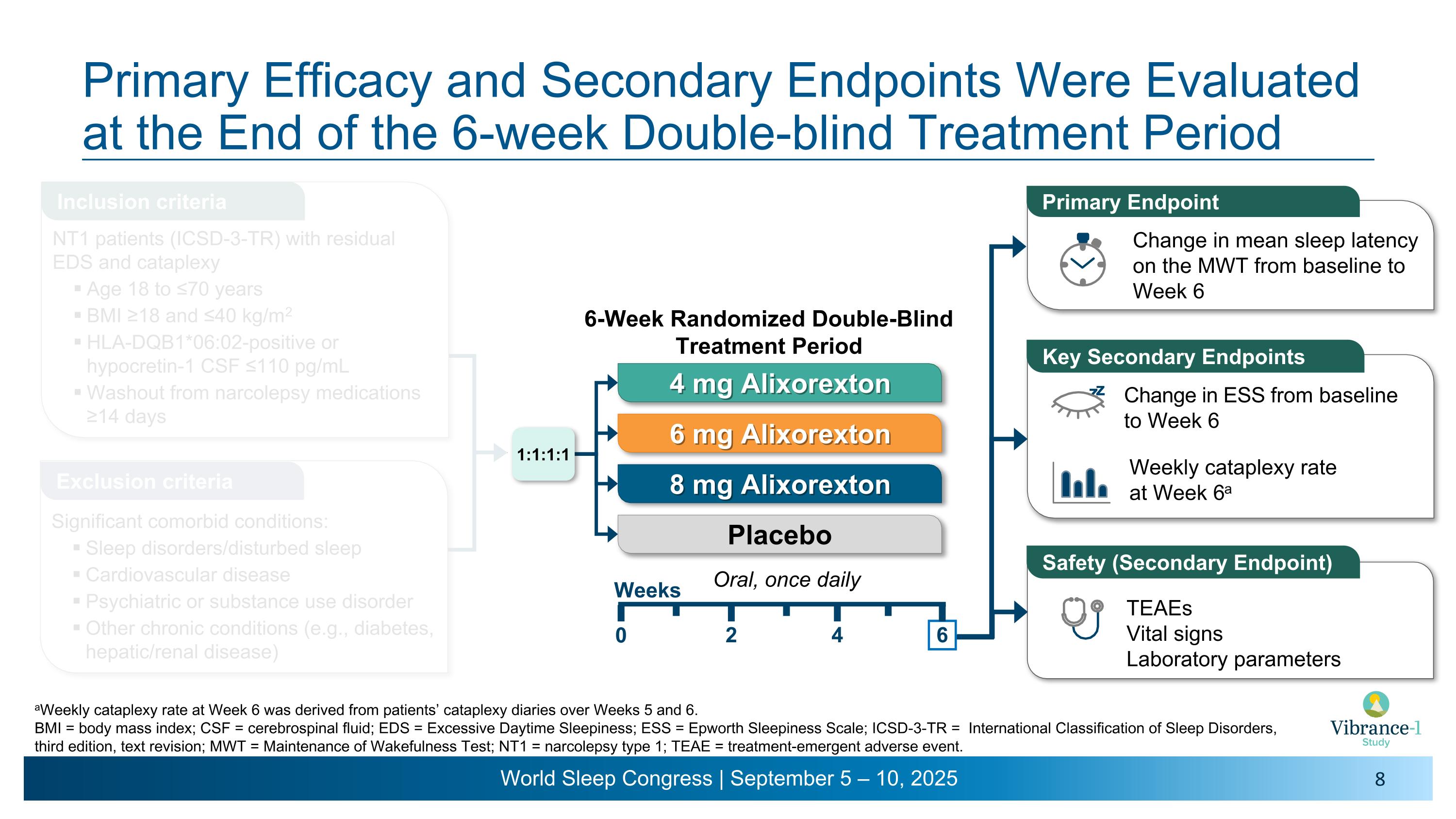
Vibrance-1 is a phase 2, randomized, double-blind, dose-range-finding, placebo-controlled study evaluating the safety and efficacy of alixorexton (formerly referred to as ALKS 2680) in adults with narcolepsy type 1 (NT1). Participants (n=92) were randomized to receive one of three doses of alixorexton (4 mg, 6 mg or 8 mg) or placebo to be taken once-daily for six weeks. The primary endpoint assessed whether participants taking alixorexton experienced an improvement in wakefulness compared to participants taking placebo, as measured by the change from baseline in mean sleep latency on the Maintenance of Wakefulness Test (MWT) at week six. Secondary endpoints included change from baseline in Epworth Sleepiness Scale (ESS) score at week six and mean weekly cataplexy rate (WCR) at weeks five and six, and incidence of adverse events. The study also included a number of exploratory patient-reported outcome measures, which evaluated the effect of alixorexton on participants’ disease severity, fatigue and cognition. All participants in the double-blind portion of the study were eligible to continue to a seven-week open-label safety extension portion of the study, followed by a long-term safety study.
About Alixorexton
Alixorexton (formerly referred to as ALKS 2680) is a novel, investigational, oral, selective orexin 2 receptor (OX2R) agonist in development as a once-daily treatment for narcolepsy type 1 (NT1), narcolepsy type 2 (NT2) and idiopathic hypersomnia (IH). Orexin, a neuropeptide produced in the lateral hypothalamus, is considered to be the master regulator of wakefulness due to its activation of multiple, downstream wake-promoting pathways that project widely throughout the brain.8 Targeting the orexin system may address excessive daytime sleepiness across hypersomnolence disorders, whether or not deficient orexin signaling is the underlying cause of disease.9 Once-daily oral administration of alixorexton was previously evaluated in a phase 1 study in healthy volunteers and patients with NT1, NT2 and IH, and is currently being evaluated in the phase 2 Vibrance-1, Vibrance-2 and Vibrance-3 studies in patients with NT1, NT2 and IH, respectively.
About Alkermes plc
Alkermes plc (Nasdaq: ALKS) is a mid-cap growth and value equity global biopharmaceutical company that seeks to develop innovative medicines in the field of neuroscience. The company has a portfolio of proprietary commercial products for the treatment of alcohol dependence, opioid dependence, schizophrenia and bipolar I disorder, and a pipeline of clinical and preclinical candidates in development for neurological disorders, including narcolepsy and idiopathic hypersomnia. Headquartered in Ireland, Alkermes also has a corporate office and research and development center in Massachusetts and a manufacturing facility in Ohio. For more information, please visit Alkermes’ website at www.alkermes.com.
Note Regarding Forward-Looking Statements
Certain statements set forth in this press release constitute “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: the potential therapeutic and commercial value of alixorexton and the company’s other orexin 2 receptor agonists, and the company’s expectations, including timelines, regarding the company’s orexin development programs. The company cautions that forward-looking statements are inherently uncertain. Although the company believes that such statements are based on reasonable assumptions within the bounds of its knowledge of its business and operations, the forward-looking statements are neither promises nor guarantees and they are necessarily subject to a high degree of uncertainty and risk. Actual performance and results may differ materially from those expressed or implied in the forward-looking statements due to various risks and uncertainties. These risks and uncertainties include, among others: whether initial clinical results for alixorexton will be predictive of results of future stages of ongoing clinical studies, future clinical studies or real-world results; whether ongoing or future clinical studies for alixorexton will be initiated or completed on expected timelines or at all; whether alixorexton could be shown to be ineffective or unsafe; potential changes in the cost, scope and duration of the alixorexton development program; and those risks and uncertainties described under the heading “Risk Factors” in the company’s Annual Report on Form 10-K for the year ended Dec. 31, 2024 and in subsequent filings made by the company with the U.S. Securities and Exchange Commission (SEC), which are available on the SEC’s website at www.sec.gov. Existing and prospective investors are cautioned not to place undue reliance on
these forward-looking statements, which speak only as of the date hereof. Except as required by law, the company disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release.
1 ESS: 8-item self-administered questionnaire that measures severity of excessive daytime sleepiness across multiple conditions over the past 7 days (≤10 = normative).
2 Week 13 data reflects data from the subset of patients who had completed the Week 13 visit of the open-label extension period as of the July 1, 2025 data snapshot (n=59). Not all patients had completed the open-label extension as of July 1, 2025.
3 Weekly cataplexy rate was derived at Week 6 from patients’ cataplexy diaries over Weeks 5 and 6.
4 NSS-CT: 15-item self-administered questionnaire (score: 0-57) that assesses the severity and consequences of the five major narcolepsy symptoms such as daytime sleepiness, cataplexy, hallucinations, sleep paralysis, and disturbed nighttime sleep over the past 7 days.
5 PROMIS-Fatigue: 6-item self-administered questionnaire assessing the severity of a patients’ fatigue over the past 7 days. Items are scored and transformed to T-scores (<55 = normative).
6 BC-CCI: 6-item self-administered questionnaire (score: 0-18) assessing perceived problems with concentration, memory, expressing thoughts, word finding, slow thinking, and difficulty solving problems over the past 7 days (≤4 = normative).
7 TEAEs in ≥10% among all alixorexton-treated patients.
8 Buysse, D. Diagnosis and assessment of sleep and circadian rhythm disorders. Journal of Psychiatric Practice. 2005; 11(2):102-115
9 Ten-Blanco M, Flores A, Cristino L, Pereda-Perez I. Targeting the orexin/hypocretin system for the treatment of neuropsychiatric and neurodegenerative diseases: From animal to clinical studies. Frontiers in Neuroendocrinology. 2023;69(101066). https://www.sciencedirect.com/science/article/pii/S0091302223000146

Vibrance-1 Alixorexton Phase 2 Study in Patients with �Narcolepsy Type 1 September 8, 2025 Exhibit 99.2

Certain statements set forth in this presentation constitute “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: the potential therapeutic and commercial value of alixorexton (formerly referred to as ALKS 2680) and the company’s expectations regarding the alixorexton development program. Actual performance and results may differ materially from those expressed or implied in the forward-looking statements due to various risks, assumptions and uncertainties. These risks, assumptions and uncertainties include, among others: whether initial clinical results for alixorexton will be predictive of results of future stages of ongoing clinical studies, future clinical studies or real-world results; whether ongoing or future clinical studies for alixorexton will be initiated or completed on expected timelines or at all; whether alixorexton could be shown to be ineffective or unsafe; potential changes in the cost, scope and duration of the alixorexton development program; and those risks, assumptions and uncertainties described under the heading “Risk Factors” in the company’s Annual Report on Form 10-K for the year ended Dec. 31, 2024 and in subsequent filings made by the company with the U.S. Securities and Exchange Commission (“SEC”), which are available on the SEC’s website at www.sec.gov, and on the company’s website at www.alkermes.com in the ‘Investors – SEC filings’ section. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. Except as required by law, the company disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this presentation. Forward-Looking Statements

Vibrance-1 Phase 2 Study of Alixorexton in Patients �With Narcolepsy Type 1 Once-daily alixorexton demonstrated new, potential best-in-class features across a range of doses Compelling therapeutic benefit; statistically significant effect on excessive daytime sleepiness and significant improvements in overall disease severity, fatigue and cognition Generally well-tolerated profile at all doses Alkermes plans to rapidly initiate global phase 3 program
Today’s Speakers Marcus Yountz, M.D.�Vice President, Clinical Development; Alixorexton Clinical Program Lead Giuseppe Plazzi, M.D., Ph.D.�Vibrance-1 Lead Investigator; Director of the Narcolepsy Center at the IRCCS of the Neurological Sciences of Bologna;�Professor of Childhood Neuropsychiatry, University of Modena and Reggio Emilia Craig Hopkinson, M.D. Executive Vice President, Chief Medical Officer Richard Pops�Chief Executive Officer

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.
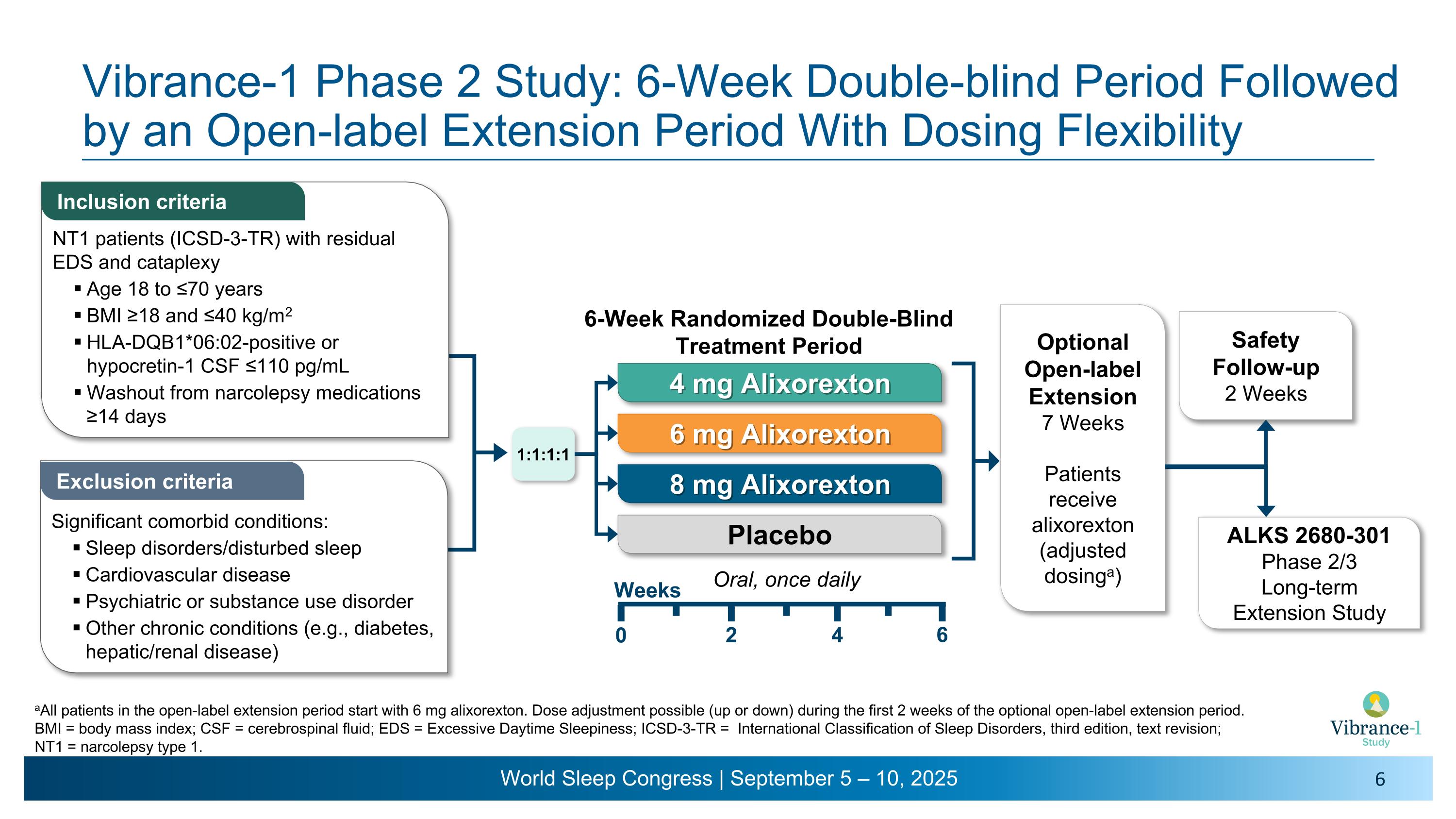
Vibrance-1 Phase 2 Study: 6-Week Double-blind Period Followed by an Open-label Extension Period With Dosing Flexibility Inclusion criteria Exclusion criteria aAll patients in the open-label extension period start with 6 mg alixorexton. Dose adjustment possible (up or down) during the first 2 weeks of the optional open-label extension period. BMI = body mass index; CSF = cerebrospinal fluid; EDS = Excessive Daytime Sleepiness; ICSD-3-TR = International Classification of Sleep Disorders, third edition, text revision; NT1 = narcolepsy type 1. NT1 patients (ICSD-3-TR) with residual EDS and cataplexy Age 18 to ≤70 years BMI ≥18 and ≤40 kg/m2 HLA-DQB1*06:02-positive or hypocretin-1 CSF ≤110 pg/mL Washout from narcolepsy medications ≥14 days Significant comorbid conditions: Sleep disorders/disturbed sleep Cardiovascular disease Psychiatric or substance use disorder Other chronic conditions (e.g., diabetes, hepatic/renal disease) Optional Open-label Extension 7 Weeks Patients receive alixorexton �(adjusted dosinga) Safety Follow-up 2 Weeks ALKS 2680-301�Phase 2/3 �Long-term �Extension Study 1:1:1:1 Oral, once daily 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo Weeks 2 4 6 0 6-Week Randomized Double-Blind Treatment Period
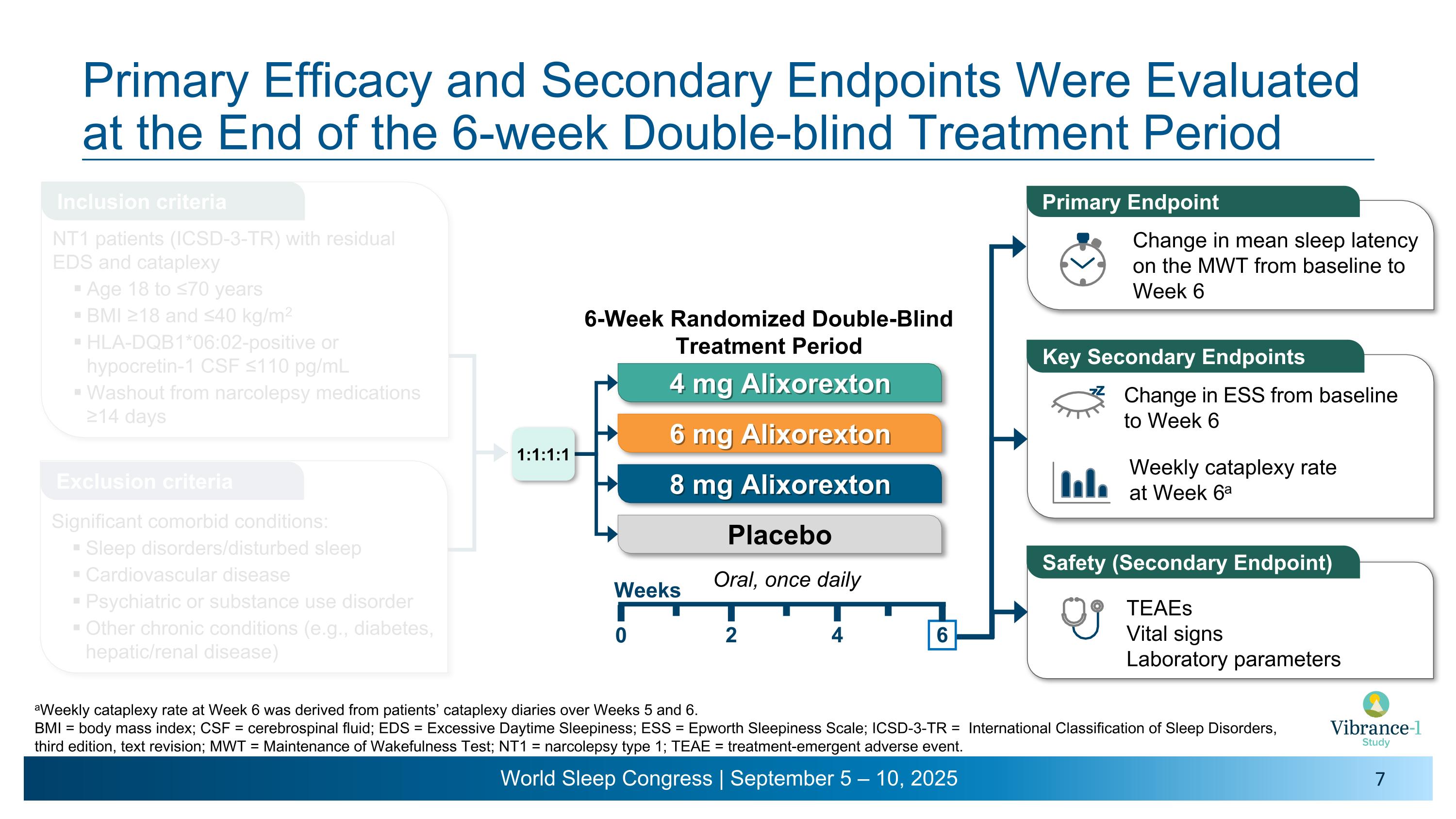
Primary Efficacy and Secondary Endpoints Were Evaluated at the End of the 6-week Double-blind Treatment Period Weekly cataplexy rate �at Week 6a Change in ESS from baseline to Week 6 Primary Endpoint Change in mean sleep latency on the MWT from baseline to Week 6 Key Secondary Endpoints z z TEAEs Vital signs Laboratory parameters Safety (Secondary Endpoint) Oral, once daily 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo 6-Week Randomized Double-Blind Treatment Period 2 4 6 0 Weeks 1:1:1:1 aWeekly cataplexy rate at Week 6 was derived from patients’ cataplexy diaries over Weeks 5 and 6.�BMI = body mass index; CSF = cerebrospinal fluid; EDS = Excessive Daytime Sleepiness; ESS = Epworth Sleepiness Scale; ICSD-3-TR = International Classification of Sleep Disorders, third edition, text revision; MWT = Maintenance of Wakefulness Test; NT1 = narcolepsy type 1; TEAE = treatment-emergent adverse event. Inclusion criteria Exclusion criteria NT1 patients (ICSD-3-TR) with residual EDS and cataplexy Age 18 to ≤70 years BMI ≥18 and ≤40 kg/m2 HLA-DQB1*06:02-positive or hypocretin-1 CSF ≤110 pg/mL Washout from narcolepsy medications ≥14 days Significant comorbid conditions: Sleep disorders/disturbed sleep Cardiovascular disease Psychiatric or substance use disorder Other chronic conditions (e.g., diabetes, hepatic/renal disease)
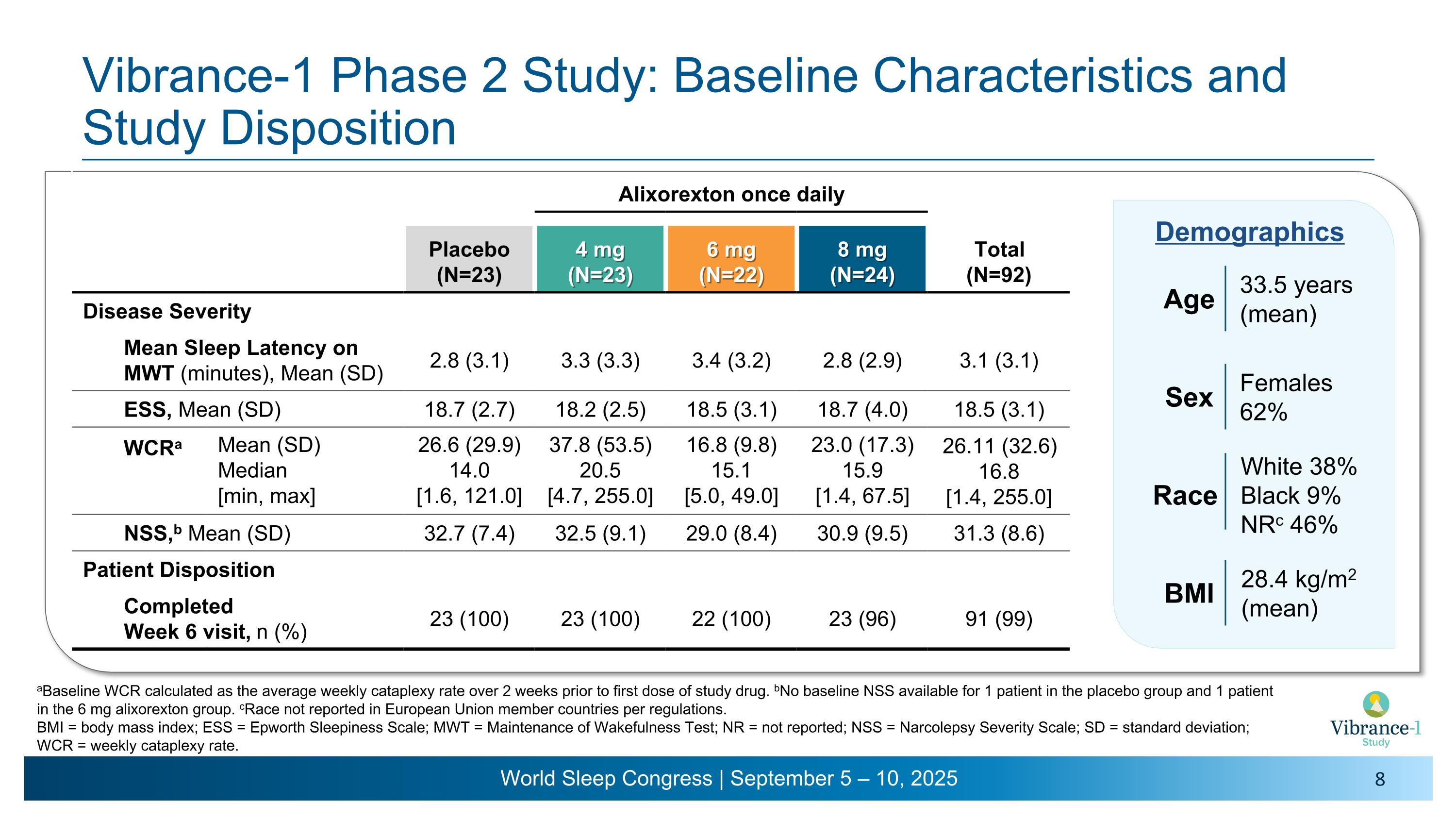
Vibrance-1 Phase 2 Study: Baseline Characteristics and Study Disposition Alixorexton once daily Placebo (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Total (N=92) Disease Severity Mean Sleep Latency on MWT (minutes), Mean (SD) 2.8 (3.1) 3.3 (3.3) 3.4 (3.2) 2.8 (2.9) 3.1 (3.1) ESS, Mean (SD) 18.7 (2.7) 18.2 (2.5) 18.5 (3.1) 18.7 (4.0) 18.5 (3.1) WCRa Mean (SD) Median [min, max] 26.6 (29.9) 14.0 �[1.6, 121.0] 37.8 (53.5) 20.5 �[4.7, 255.0] 16.8 (9.8) 15.1 �[5.0, 49.0] 23.0 (17.3) 15.9 �[1.4, 67.5] 26.11 (32.6) 16.8 [1.4, 255.0] NSS,b Mean (SD) 32.7 (7.4) 32.5 (9.1) 29.0 (8.4) 30.9 (9.5) 31.3 (8.6) Patient Disposition Completed �Week 6 visit, n (%) 23 (100) 23 (100) 22 (100) 23 (96) 91 (99) 33.5 years (mean) Age Females 62% Sex Race White 38% Black 9% NRc 46% 28.4 kg/m2 �(mean) BMI Demographics aBaseline WCR calculated as the average weekly cataplexy rate over 2 weeks prior to first dose of study drug. bNo baseline NSS available for 1 patient in the placebo group and 1 patient in the 6 mg alixorexton group. cRace not reported in European Union member countries per regulations. BMI = body mass index; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NR = not reported; NSS = Narcolepsy Severity Scale; SD = standard deviation; WCR = weekly cataplexy rate.
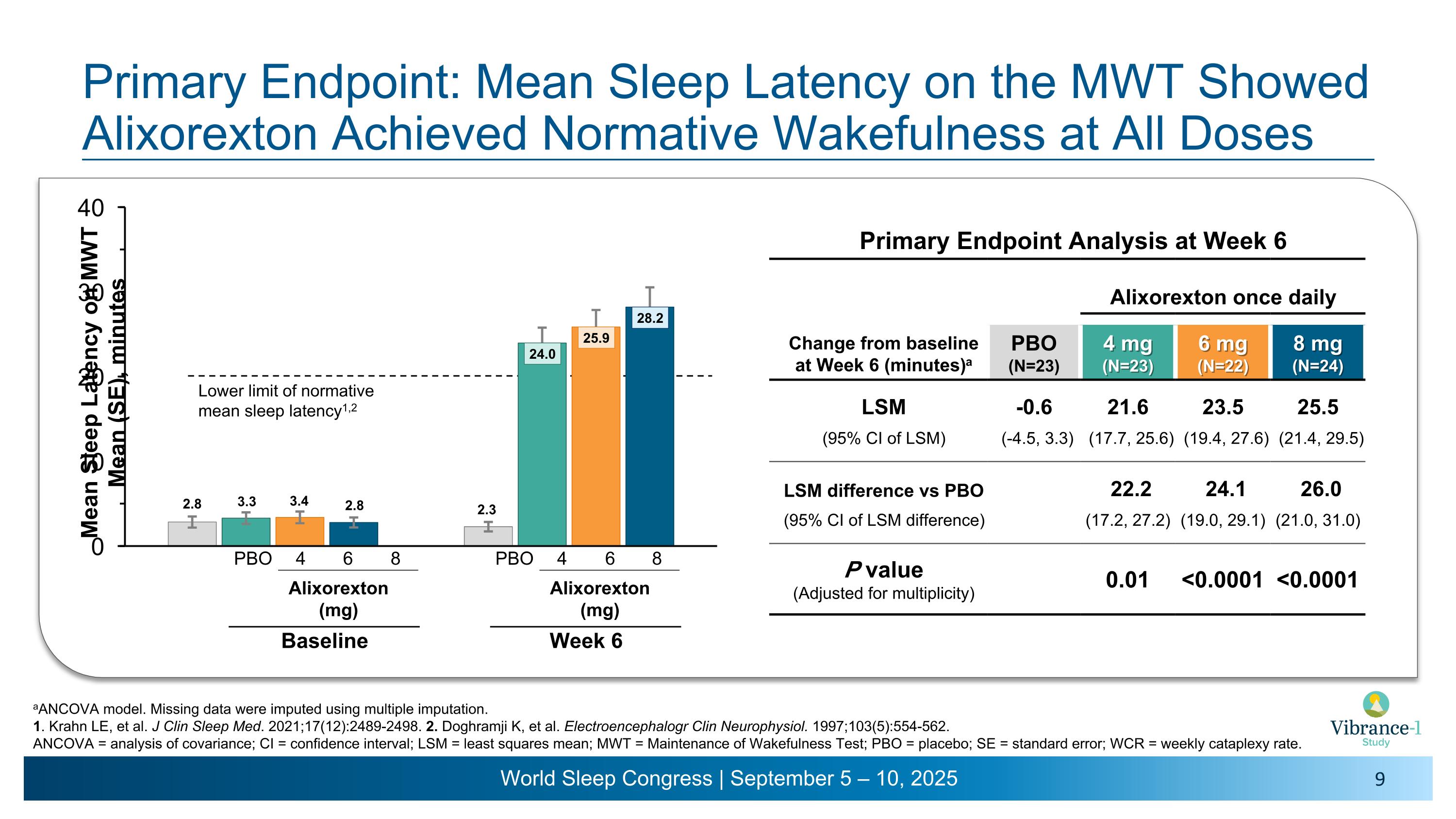
Primary Endpoint: Mean Sleep Latency on the MWT Showed Alixorexton Achieved Normative Wakefulness at All Doses Primary Endpoint Analysis at Week 6 Alixorexton once daily Change from baseline �at Week 6 (minutes)a PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) LSM -0.6 21.6 23.5 25.5 (95% CI of LSM) (-4.5, 3.3) (17.7, 25.6) (19.4, 27.6) (21.4, 29.5) LSM difference vs PBO 22.2 24.1 26.0 (95% CI of LSM difference) (17.2, 27.2) (19.0, 29.1) (21.0, 31.0) P value (Adjusted for multiplicity) 0.01 <0.0001 <0.0001 aANCOVA model. Missing data were imputed using multiple imputation. 1. Krahn LE, et al. J Clin Sleep Med. 2021;17(12):2489-2498. 2. Doghramji K, et al. Electroencephalogr Clin Neurophysiol. 1997;103(5):554-562. ANCOVA = analysis of covariance; CI = confidence interval; LSM = least squares mean; MWT = Maintenance of Wakefulness Test; PBO = placebo; SE = standard error; WCR = weekly cataplexy rate. Lower limit of normative mean sleep latency1,2 4 6 8 PBO Alixorexton �(mg) Baseline 4 6 8 PBO Alixorexton �(mg) Week 6
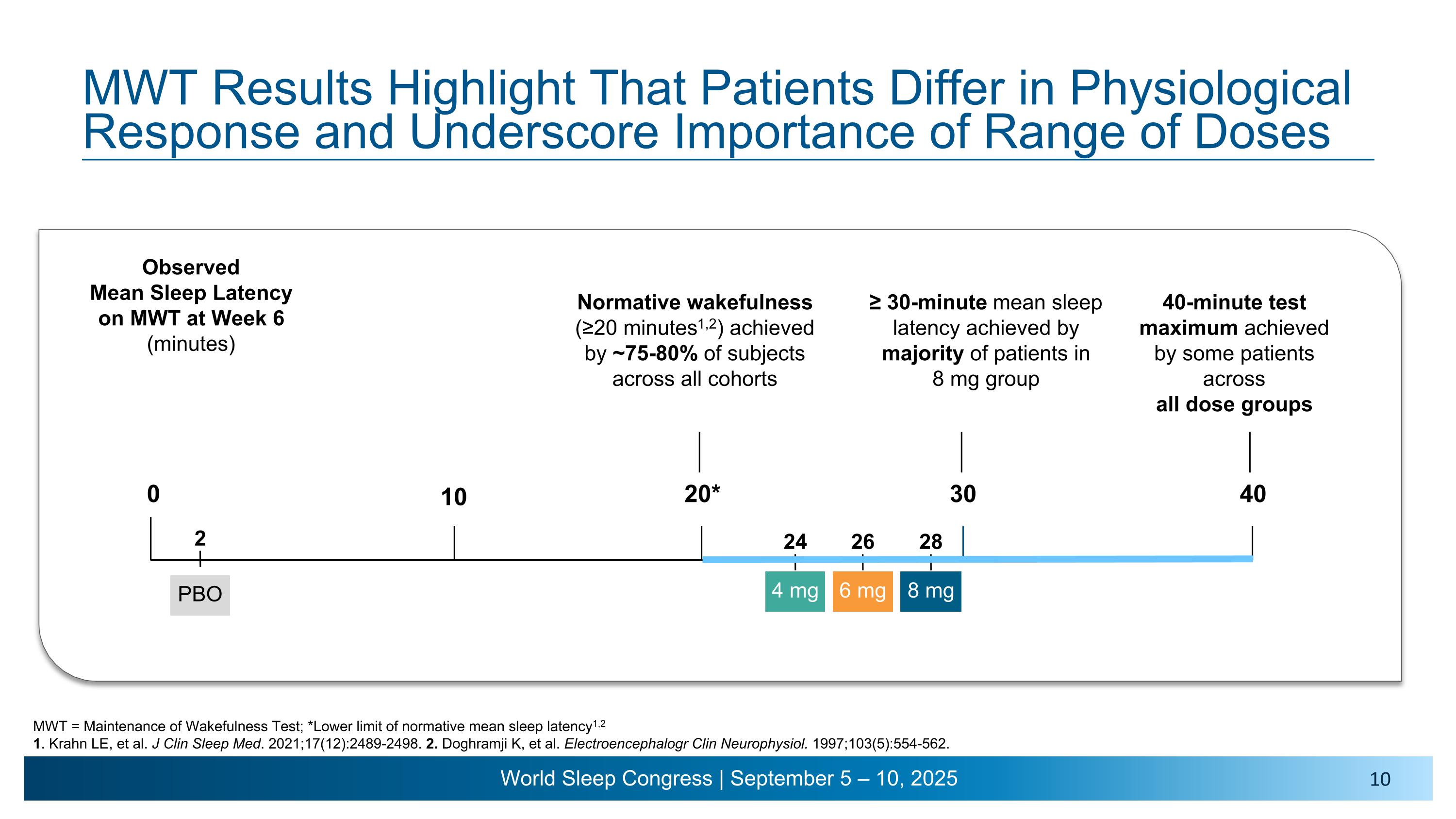
MWT Results Highlight That Patients Differ in Physiological Response and Underscore Importance of Range of Doses Normative wakefulness �(≥20 minutes1,2) achieved by ~75-80% of subjects across all cohorts ≥ 30-minute mean sleep latency achieved by majority of patients in �8 mg group 40-minute test maximum achieved by some patients across �all dose groups Observed �Mean Sleep Latency �on MWT at Week 6�(minutes) 0 20* 30 40 10 4 mg 24 26 28 6 mg 8 mg 2 PBO MWT = Maintenance of Wakefulness Test; *Lower limit of normative mean sleep latency1,2 1. Krahn LE, et al. J Clin Sleep Med. 2021;17(12):2489-2498. 2. Doghramji K, et al. Electroencephalogr Clin Neurophysiol. 1997;103(5):554-562.
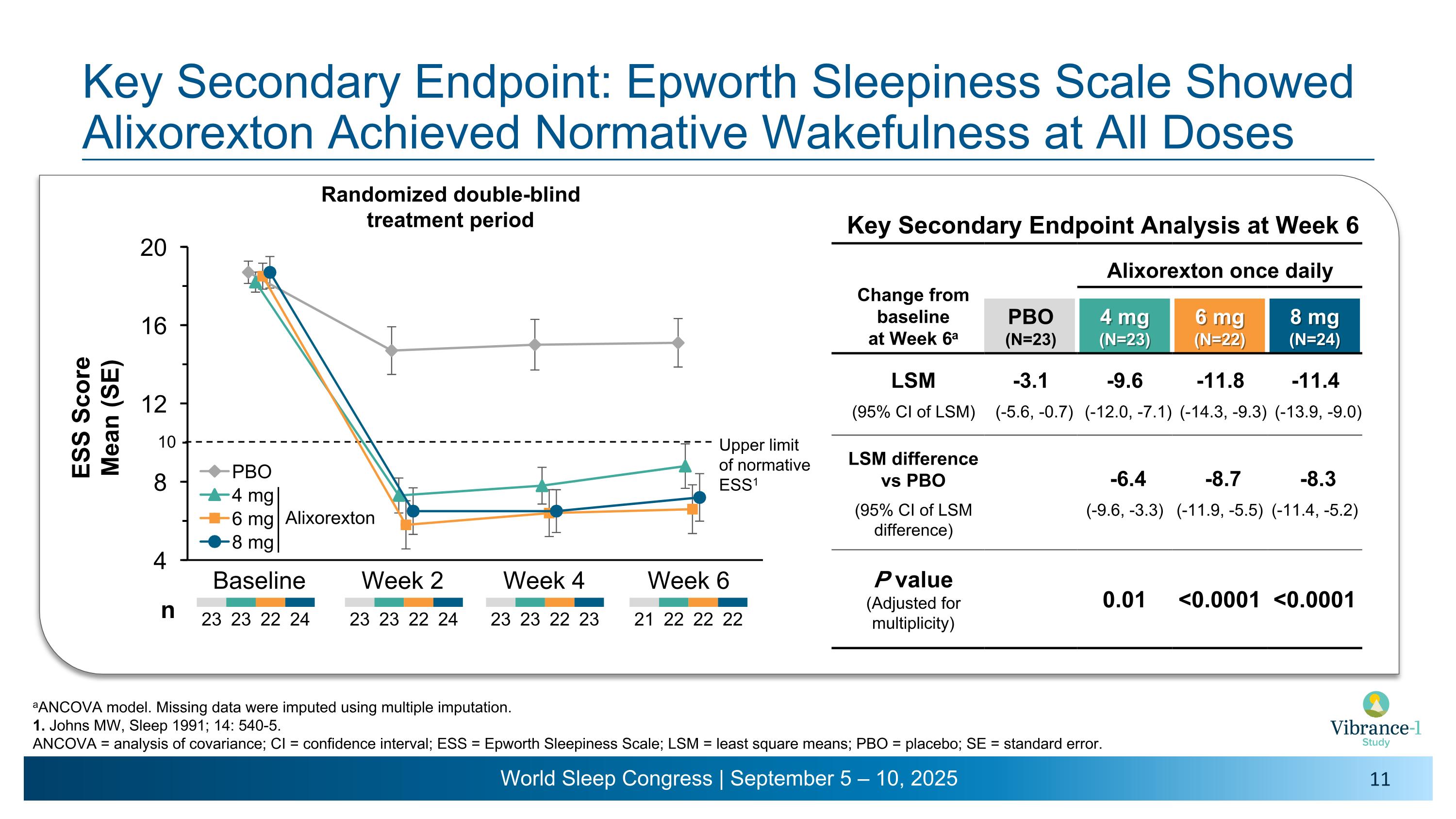
Key Secondary Endpoint: Epworth Sleepiness Scale Showed Alixorexton Achieved Normative Wakefulness at All Doses aANCOVA model. Missing data were imputed using multiple imputation. 1. Johns MW, Sleep 1991; 14: 540-5. ANCOVA = analysis of covariance; CI = confidence interval; ESS = Epworth Sleepiness Scale; LSM = least square means; PBO = placebo; SE = standard error. Baseline Week 2 Week 4 Week 6 23 23 22 24 23 23 22 23 23 23 22 24 n Randomized double-blind �treatment period Alixorexton 21 22 22 22 Key Secondary Endpoint Analysis at Week 6 Change from �baseline �at Week 6a Alixorexton once daily PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) LSM -3.1 -9.6 -11.8 -11.4 (95% CI of LSM) (-5.6, -0.7) (-12.0, -7.1) (-14.3, -9.3) (-13.9, -9.0) LSM difference vs PBO -6.4 -8.7 -8.3 (95% CI of LSM difference) (-9.6, -3.3) (-11.9, -5.5) (-11.4, -5.2) P value (Adjusted for multiplicity) 0.01 <0.0001 <0.0001 Upper limit �of normative ESS1 10
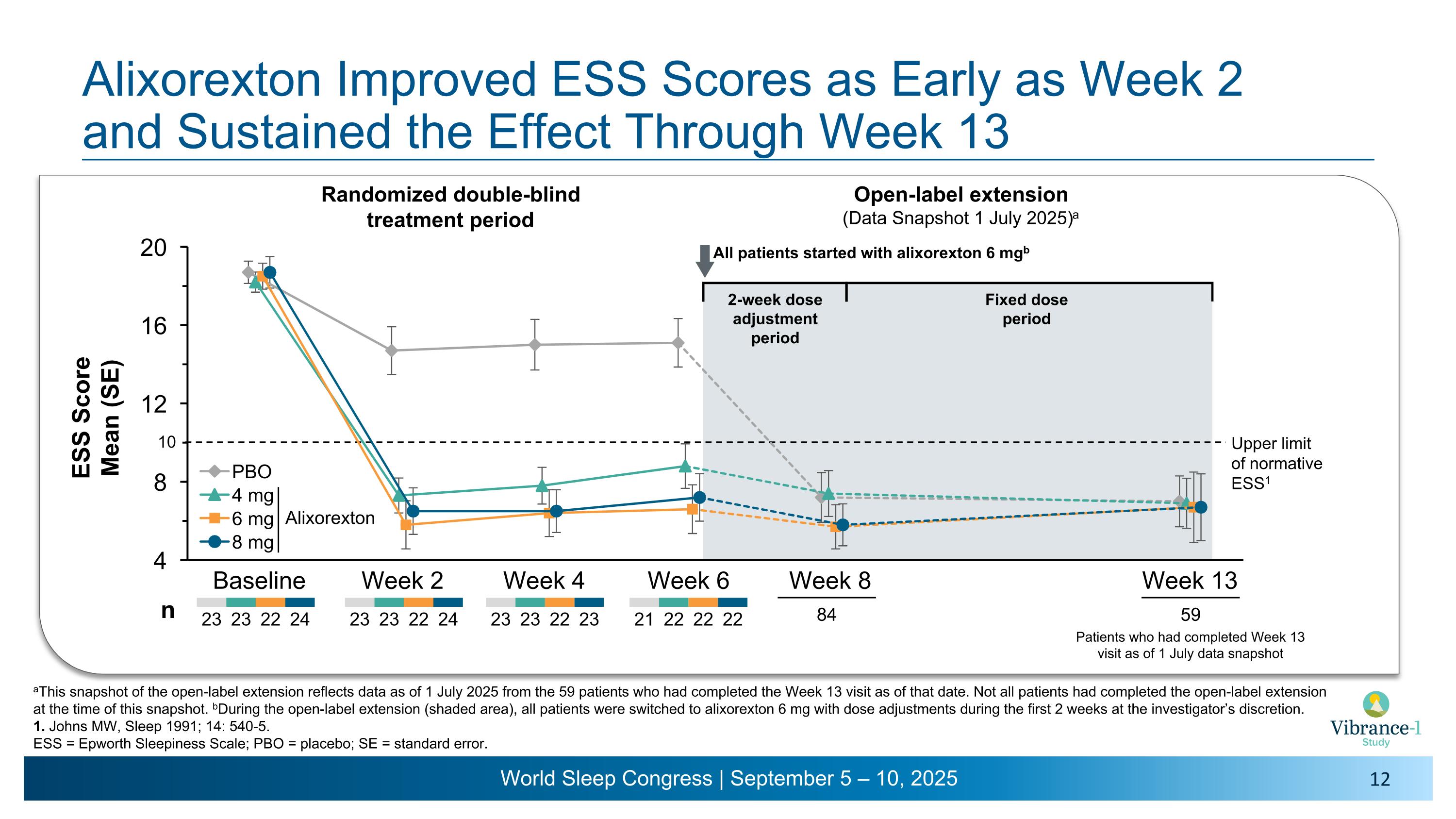
Alixorexton Improved ESS Scores as Early as Week 2 �and Sustained the Effect Through Week 13 aThis snapshot of the open-label extension reflects data as of 1 July 2025 from the 59 patients who had completed the Week 13 visit as of that date. Not all patients had completed the open-label extension at the time of this snapshot. bDuring the open-label extension (shaded area), all patients were switched to alixorexton 6 mg with dose adjustments during the first 2 weeks at the investigator’s discretion. 1. Johns MW, Sleep 1991; 14: 540-5. ESS = Epworth Sleepiness Scale; PBO = placebo; SE = standard error. Baseline Week 2 Week 4 Week 6 Week 8 Upper limit �of normative ESS1 Week 13 23 23 22 24 23 23 22 23 23 23 22 24 84 All patients started with alixorexton 6 mgb n Open-label extension (Data Snapshot 1 July 2025)a Randomized double-blind �treatment period 21 22 22 22 Patients who had completed Week 13 visit as of 1 July data snapshot 59 2-week dose �adjustment period Fixed dose period 10 Alixorexton
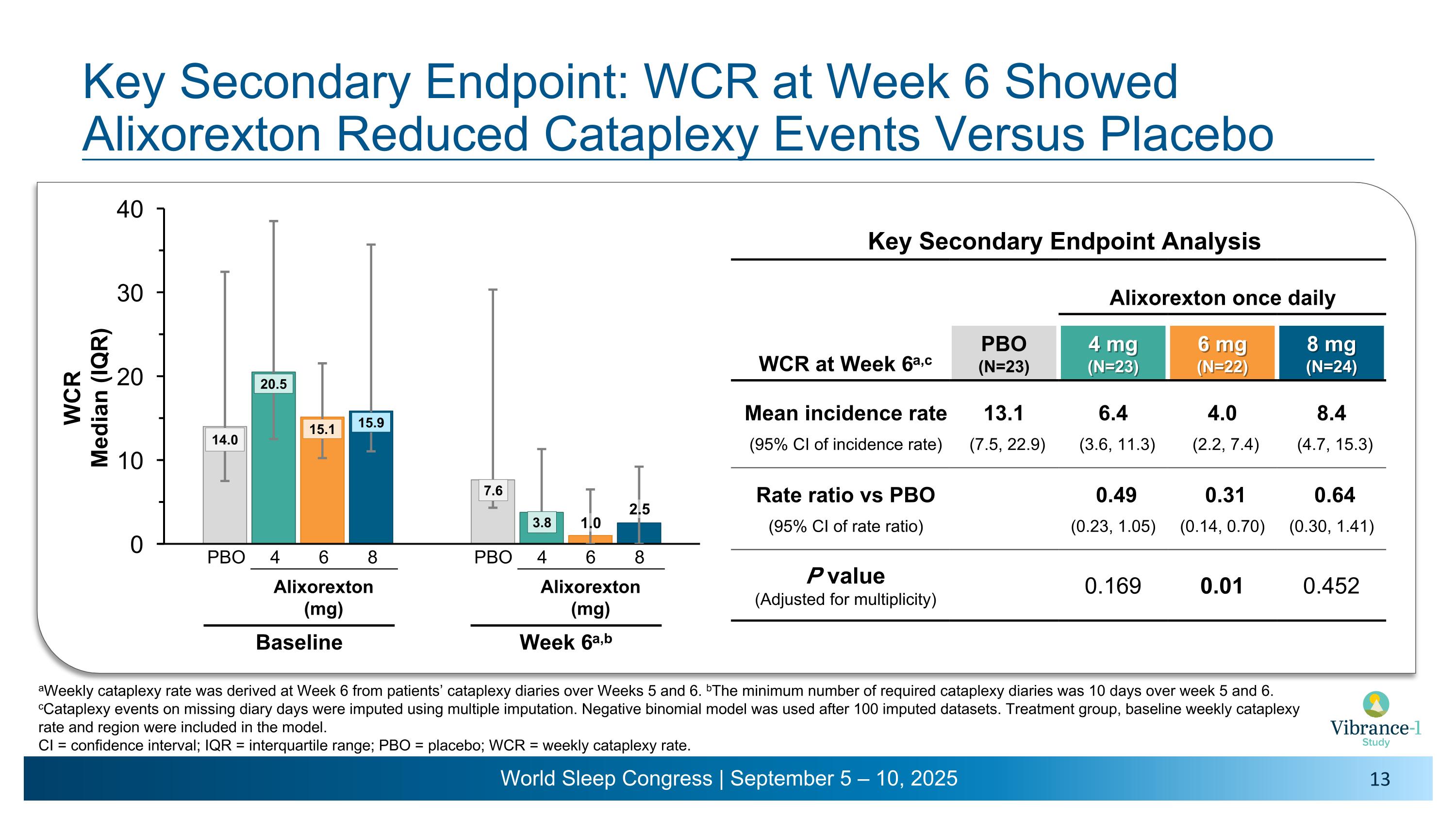
Key Secondary Endpoint: WCR at Week 6 Showed �Alixorexton Reduced Cataplexy Events Versus Placebo Key Secondary Endpoint Analysis Alixorexton once daily WCR at Week 6a,c PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) Mean incidence rate 13.1 6.4 4.0 8.4 (95% CI of incidence rate) (7.5, 22.9) (3.6, 11.3) (2.2, 7.4) (4.7, 15.3) Rate ratio vs PBO 0.49 0.31 0.64 (95% CI of rate ratio) (0.23, 1.05) (0.14, 0.70) (0.30, 1.41) P value (Adjusted for multiplicity) 0.169 0.01 0.452 aWeekly cataplexy rate was derived at Week 6 from patients’ cataplexy diaries over Weeks 5 and 6. bThe minimum number of required cataplexy diaries was 10 days over week 5 and 6. cCataplexy events on missing diary days were imputed using multiple imputation. Negative binomial model was used after 100 imputed datasets. Treatment group, baseline weekly cataplexy rate and region were included in the model. �CI = confidence interval; IQR = interquartile range; PBO = placebo; WCR = weekly cataplexy rate. 1.0 2.5 4 6 8 PBO Alixorexton �(mg) Baseline 4 6 8 PBO Alixorexton �(mg) Week 6a,b
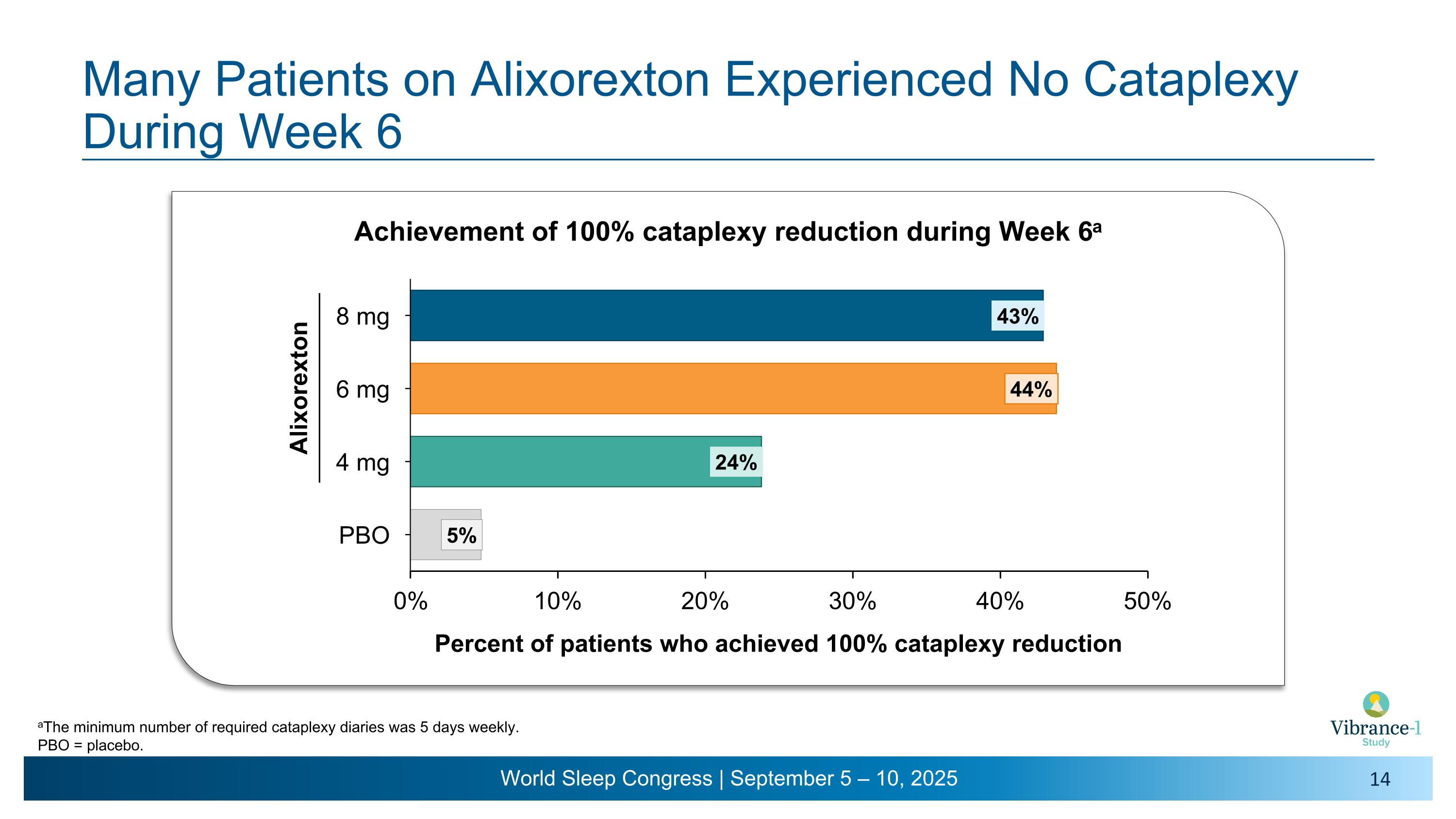
Many Patients on Alixorexton Experienced No Cataplexy During Week 6 = Alixorexton Achievement of 100% cataplexy reduction during Week 6a aThe minimum number of required cataplexy diaries was 5 days weekly. PBO = placebo.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.
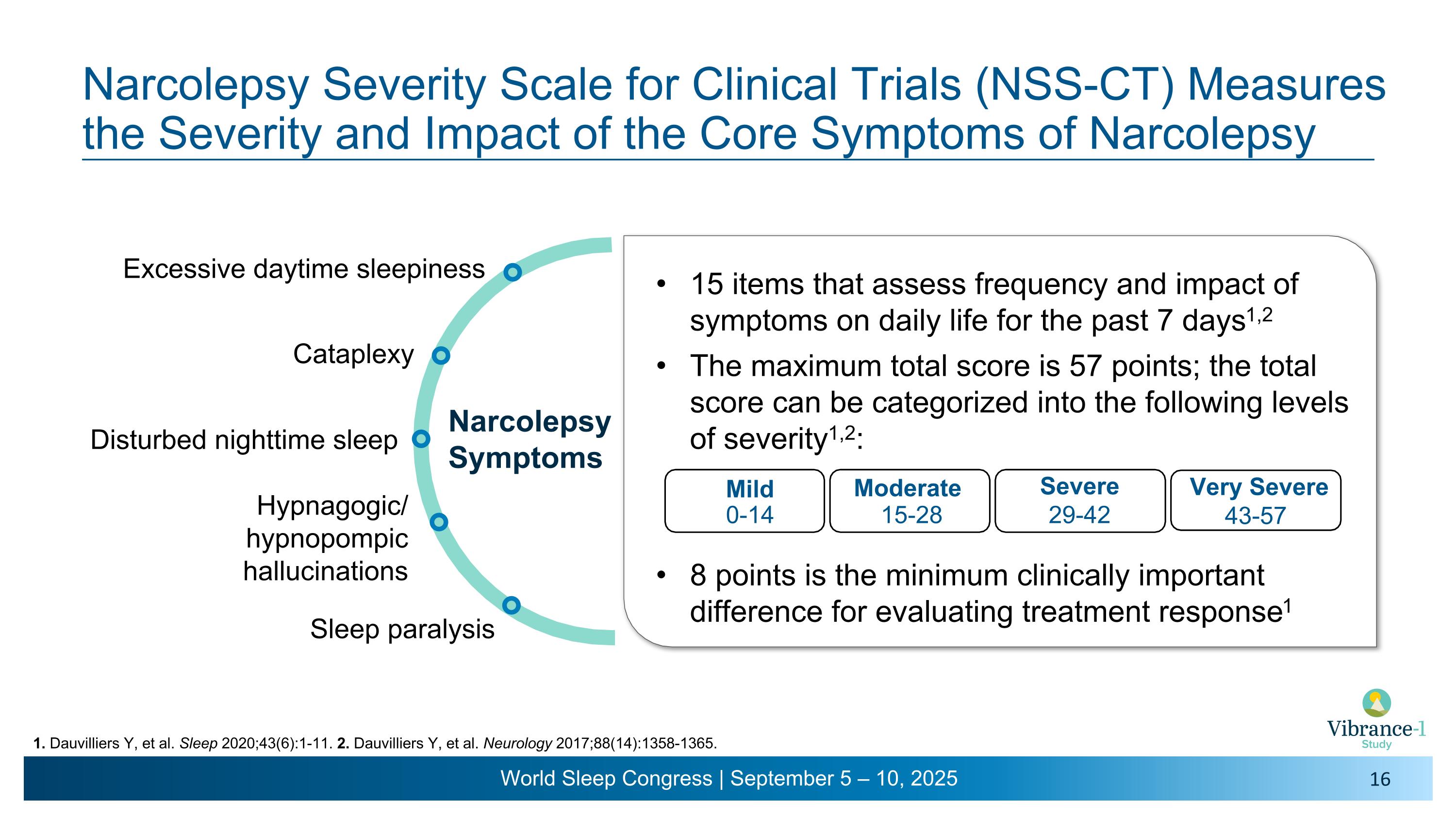
Narcolepsy Symptoms Excessive daytime sleepiness Cataplexy Hypnagogic/�hypnopompic hallucinations Sleep paralysis Narcolepsy Severity Scale for Clinical Trials (NSS-CT) Measures the Severity and Impact of the Core Symptoms of Narcolepsy 1. Dauvilliers Y, et al. Sleep 2020;43(6):1-11. 2. Dauvilliers Y, et al. Neurology 2017;88(14):1358-1365. Disturbed nighttime sleep 15 items that assess frequency and impact of symptoms on daily life for the past 7 days1,2 The maximum total score is 57 points; the total score can be categorized into the following levels of severity1,2: 8 points is the minimum clinically important difference for evaluating treatment response1 Mild Moderate Severe 0-14 15-28 29-42 Very Severe 43-57
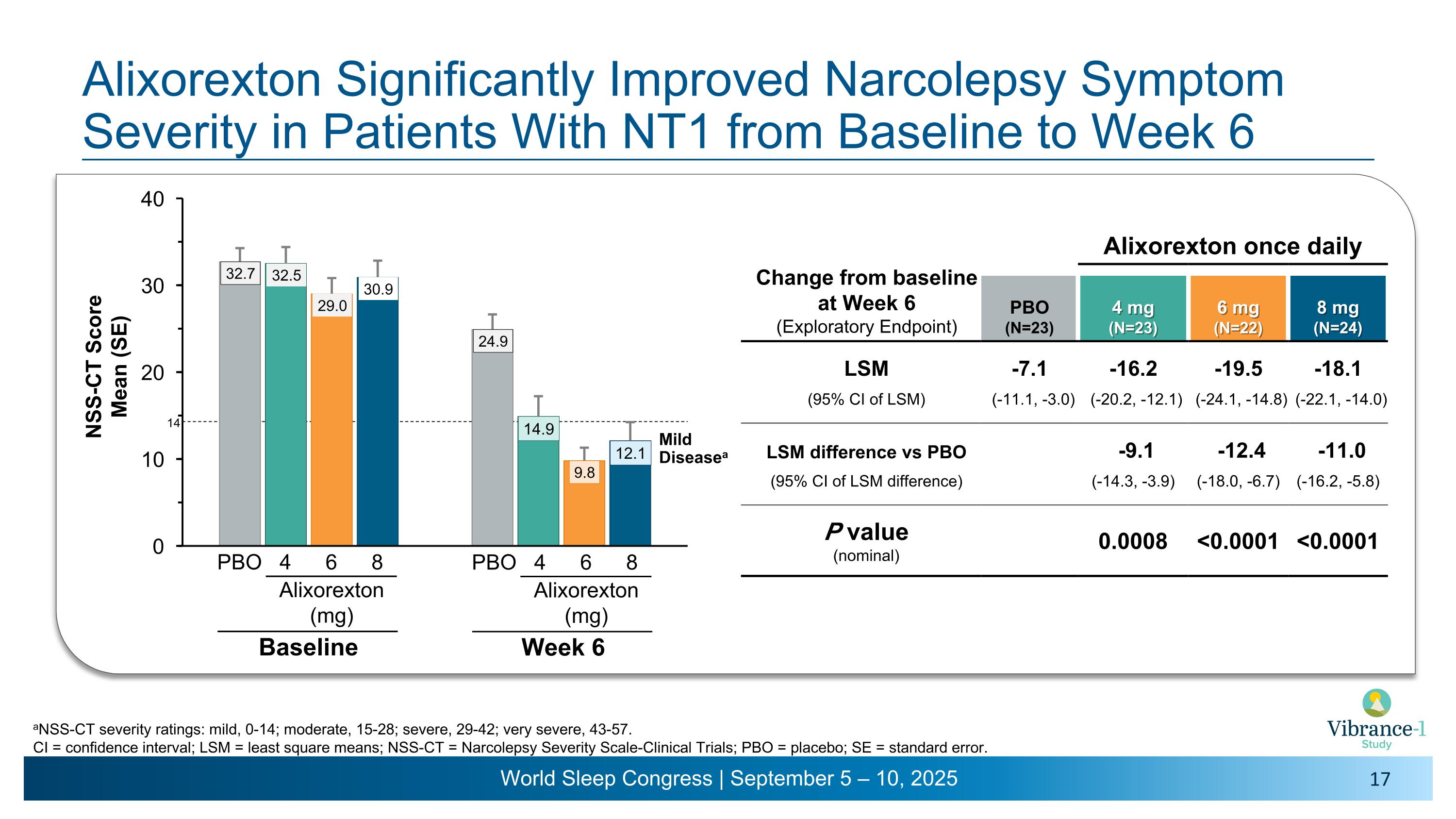
Alixorexton Significantly Improved Narcolepsy Symptom Severity in Patients With NT1 from Baseline to Week 6 aNSS-CT severity ratings: mild, 0-14; moderate, 15-28; severe, 29-42; very severe, 43-57. CI = confidence interval; LSM = least square means; NSS-CT = Narcolepsy Severity Scale-Clinical Trials; PBO = placebo; SE = standard error. Change from baseline �at Week 6 �(Exploratory Endpoint) Alixorexton once daily PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) LSM -7.1 -16.2 -19.5 -18.1 (95% CI of LSM) (-11.1, -3.0) (-20.2, -12.1) (-24.1, -14.8) (-22.1, -14.0) LSM difference vs PBO -9.1 -12.4 -11.0 (95% CI of LSM difference) (-14.3, -3.9) (-18.0, -6.7) (-16.2, -5.8) P value (nominal) 0.0008 <0.0001 <0.0001 4 6 8 PBO Alixorexton �(mg) Baseline Mild Diseasea 14 4 6 8 PBO Alixorexton �(mg) Week 6
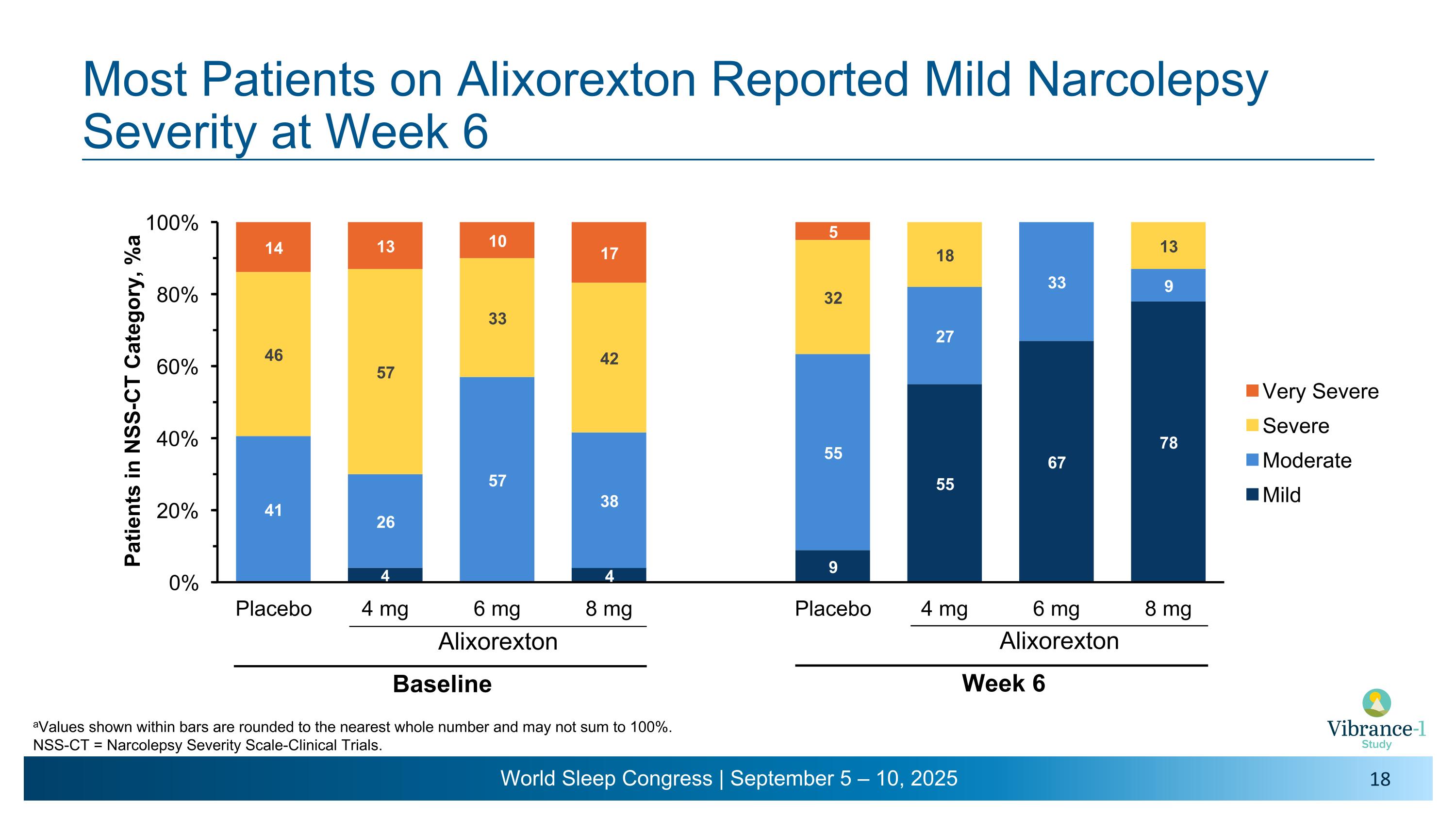
Most Patients on Alixorexton Reported Mild Narcolepsy Severity at Week 6 Alixorexton Baseline aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. NSS-CT = Narcolepsy Severity Scale-Clinical Trials. Alixorexton Week 6

Fatigue Is a Persistent, Severe, and Debilitating Unmet Need That Impacts the Lives of Patients With Narcolepsy Most narcolepsy patients experience fatigue, which often persists even with treatment1-3 Fatigue negatively affects patients’ mental health, functional outcomes and overall quality of life4 PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System.�1. AASM 2023. International Classification of Sleep Disorders – Third Edition: Text Revision, Darien, IL. 2. Maski K, et al. J Clin Sleep Med 2017;13(3):419-425. 3. Doane M, et al. Poster presented at Psych Congress 2023. 4. Droogleever F, et al. Sleep, 21(2), 163-169. PROMIS-Fatigue 6a Short Form A 6-item questionnaire scored on a 5-point Likert scale assessing the severity of fatigue in the past 7 days Items are scored and transformed to T-scores, ranging from 33.4 to 76.8 Scores less than 55 are considered normal, while scores ≥70 are considered severe A single item assessing patient-reported severity of fatigue over the past 7 days with scale responses of none, mild, moderate, severe, or very severe PGI-S Fatigue Commonly Used and Established Patient-reported Measures Specific to Fatigue
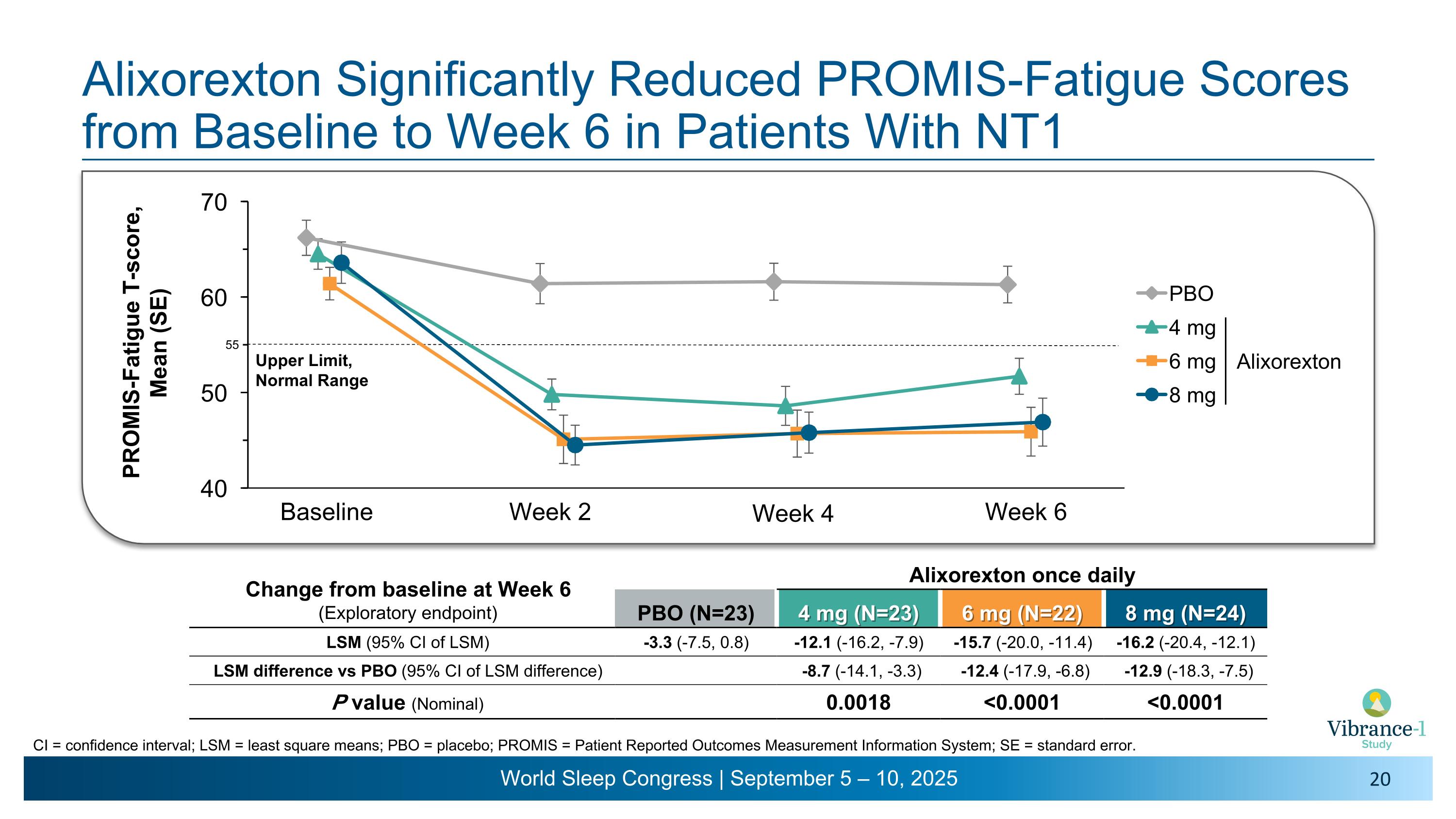
Alixorexton Significantly Reduced PROMIS-Fatigue Scores from Baseline to Week 6 in Patients With NT1 Baseline Week 2 Week 4 Week 6 CI = confidence interval; LSM = least square means; PBO = placebo; PROMIS = Patient Reported Outcomes Measurement Information System; SE = standard error. Upper Limit, Normal Range Change from baseline at Week 6 (Exploratory endpoint) Alixorexton once daily PBO (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) LSM (95% CI of LSM) -3.3 (-7.5, 0.8) -12.1 (-16.2, -7.9) -15.7 (-20.0, -11.4) -16.2 (-20.4, -12.1) LSM difference vs PBO (95% CI of LSM difference) -8.7 (-14.1, -3.3) -12.4 (-17.9, -6.8) -12.9 (-18.3, -7.5) P value (Nominal) 0.0018 <0.0001 <0.0001 55 Alixorexton
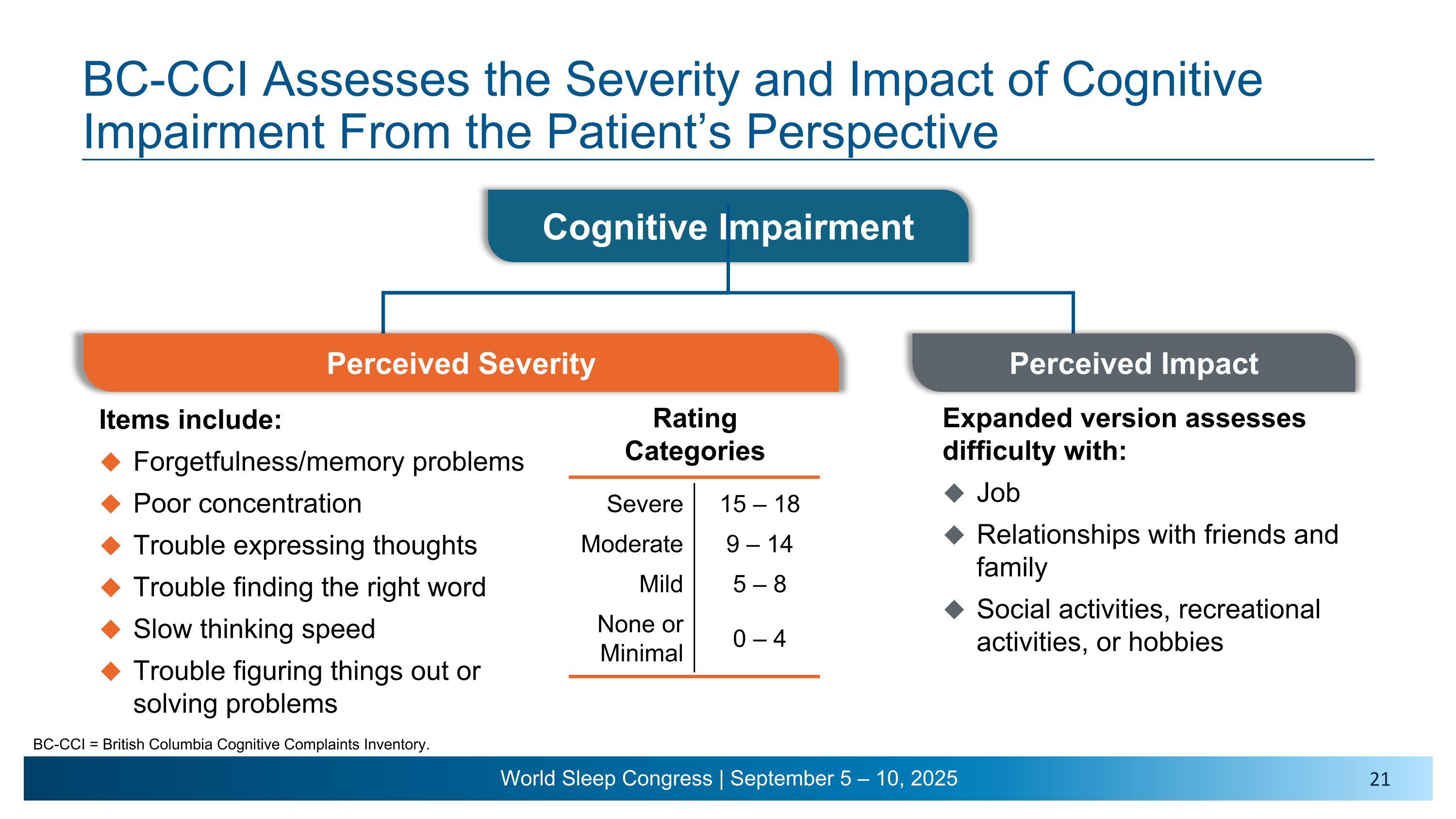
Cognitive Impairment BC-CCI Assesses the Severity and Impact of Cognitive Impairment From the Patient’s Perspective BC-CCI = British Columbia Cognitive Complaints Inventory. Items include: Forgetfulness/memory problems Poor concentration Trouble expressing thoughts Trouble finding the right word Slow thinking speed Trouble figuring things out or solving problems Rating Categories Severe 15 – 18 Moderate 9 – 14 Mild 5 – 8 None or Minimal 0 – 4 Perceived Impact Expanded version assesses difficulty with: Job Relationships with friends and family Social activities, recreational activities, or hobbies Perceived Severity
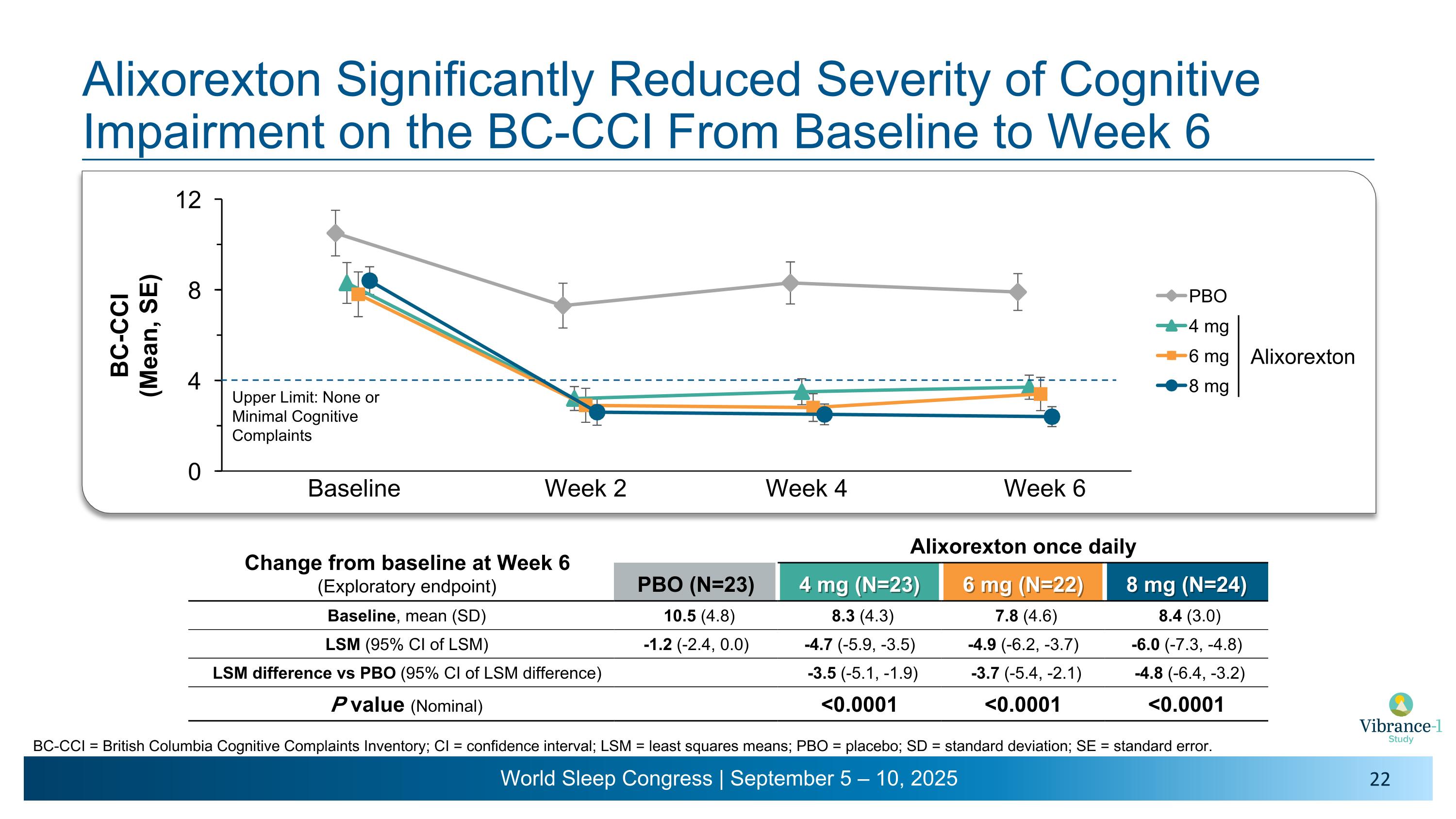
Baseline Week 2 Week 4 Week 6 Alixorexton Significantly Reduced Severity of Cognitive Impairment on the BC-CCI From Baseline to Week 6 BC-CCI = British Columbia Cognitive Complaints Inventory; CI = confidence interval; LSM = least squares means; PBO = placebo; SD = standard deviation; SE = standard error. Upper Limit: None or Minimal Cognitive Complaints Change from baseline at Week 6 (Exploratory endpoint) Alixorexton once daily PBO (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Baseline, mean (SD) 10.5 (4.8) 8.3 (4.3) 7.8 (4.6) 8.4 (3.0) LSM (95% CI of LSM) -1.2 (-2.4, 0.0) -4.7 (-5.9, -3.5) -4.9 (-6.2, -3.7) -6.0 (-7.3, -4.8) LSM difference vs PBO (95% CI of LSM difference) -3.5 (-5.1, -1.9) -3.7 (-5.4, -2.1) -4.8 (-6.4, -3.2) P value (Nominal) <0.0001 <0.0001 <0.0001 Alixorexton
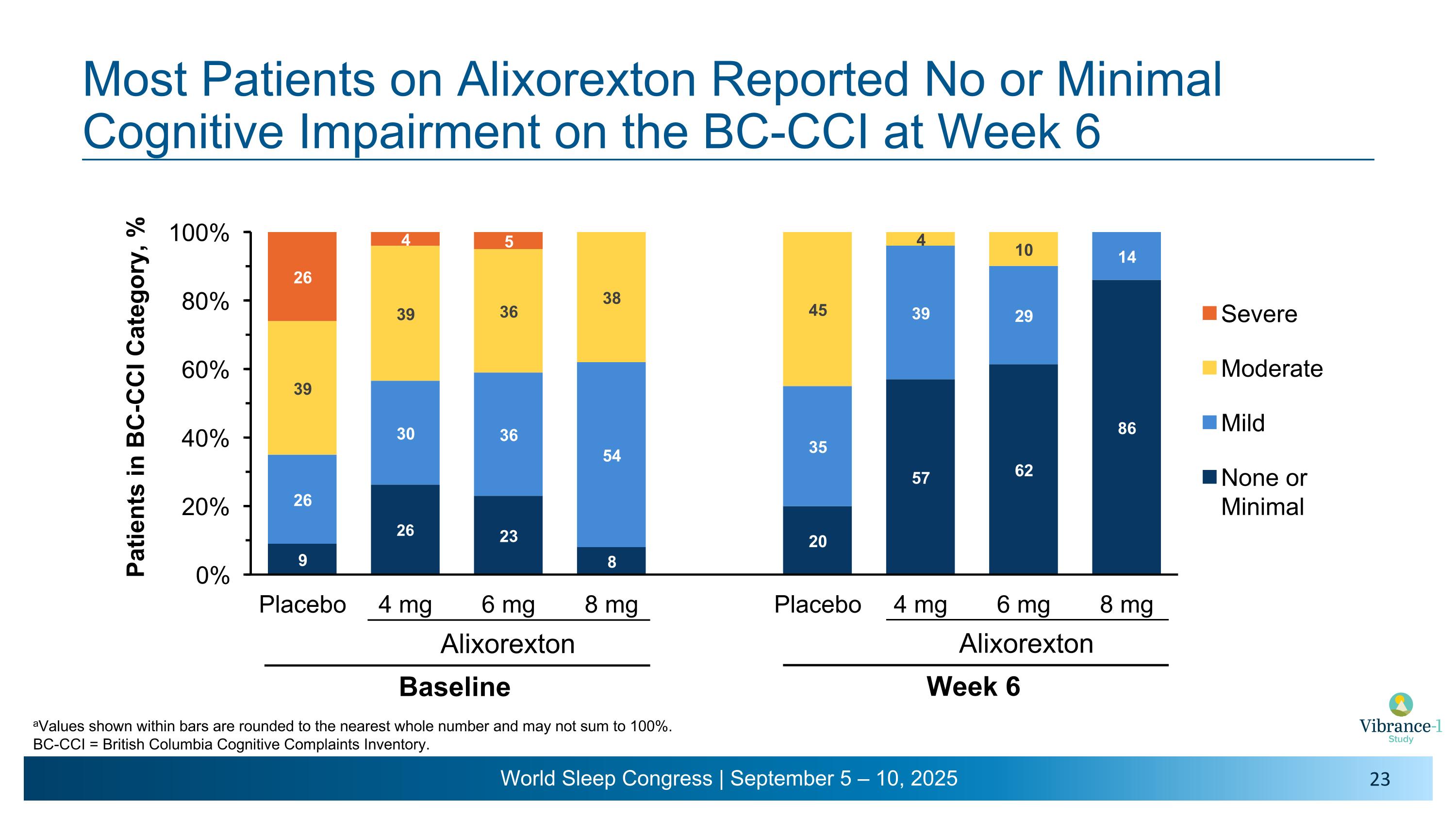
Most Patients on Alixorexton Reported No or Minimal Cognitive Impairment on the BC-CCI at Week 6 Alixorexton Baseline Alixorexton Week 6 aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. BC-CCI = British Columbia Cognitive Complaints Inventory.
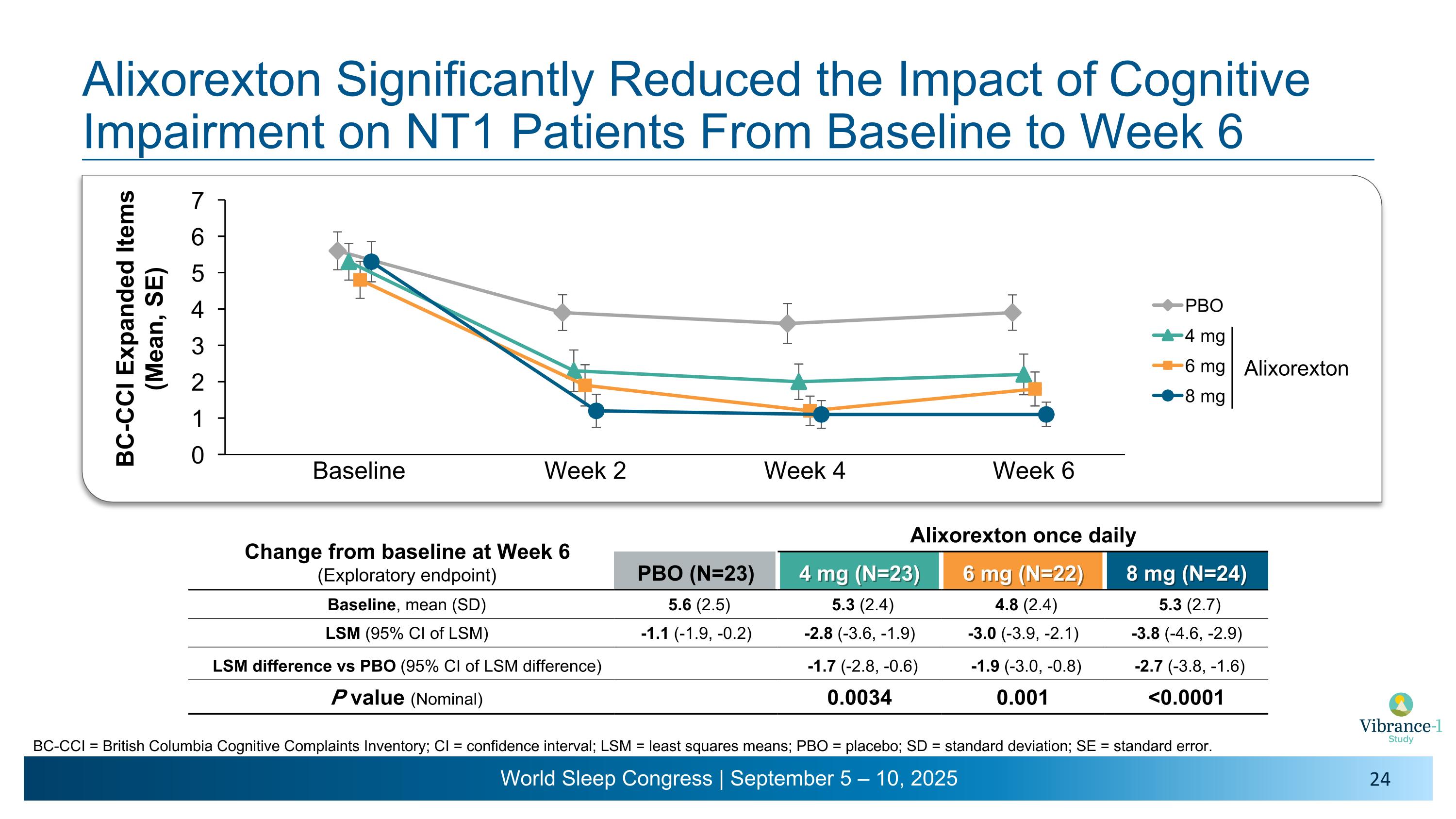
Alixorexton Significantly Reduced the Impact of Cognitive Impairment on NT1 Patients From Baseline to Week 6 Baseline Week 2 Week 4 Week 6 Change from baseline at Week 6�(Exploratory endpoint) Alixorexton once daily PBO (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Baseline, mean (SD) 5.6 (2.5) 5.3 (2.4) 4.8 (2.4) 5.3 (2.7) LSM (95% CI of LSM) -1.1 (-1.9, -0.2) -2.8 (-3.6, -1.9) -3.0 (-3.9, -2.1) -3.8 (-4.6, -2.9) LSM difference vs PBO (95% CI of LSM difference) -1.7 (-2.8, -0.6) -1.9 (-3.0, -0.8) -2.7 (-3.8, -1.6) P value (Nominal) 0.0034 0.001 <0.0001 Alixorexton BC-CCI = British Columbia Cognitive Complaints Inventory; CI = confidence interval; LSM = least squares means; PBO = placebo; SD = standard deviation; SE = standard error.

Patient-reported Outcome Measures Demonstrated Robustness and Consistency Across Doses and Assays Alixorexton is the first once-daily orexin 2 receptor agonist to demonstrate normalization of cognitive functioning and fatigue scores on patient-reported scales in addition to clinically meaningful improvements in severity of symptoms �in the NT1 population Narcolepsy Severity Scale (NSS-CT) Clinical Global Impression (CGI) – Severity Patient Global Impression (PGI) – Severity PROMIS-Fatigue 6a Short Form PGI-S Fatigue British Columbia Cognitive Complaints Inventory (BC-CCI) BC-CCI Expanded PGI-S Cognition
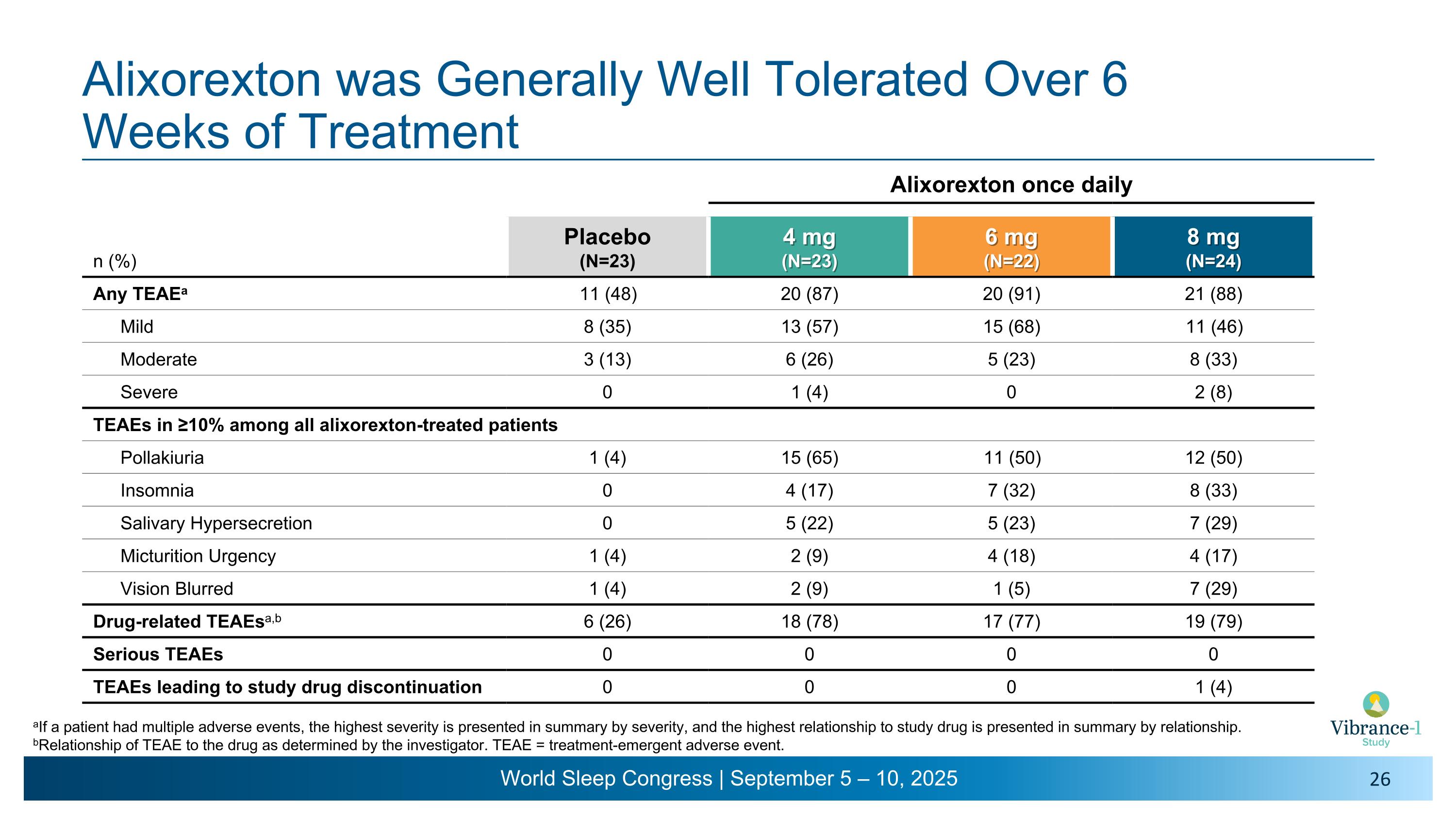
Alixorexton was Generally Well Tolerated Over 6 �Weeks of Treatment aIf a patient had multiple adverse events, the highest severity is presented in summary by severity, and the highest relationship to study drug is presented in summary by relationship. bRelationship of TEAE to the drug as determined by the investigator. TEAE = treatment-emergent adverse event. Alixorexton once daily n (%) Placebo (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Any TEAEa 11 (48) 20 (87) 20 (91) 21 (88) Mild 8 (35) 13 (57) 15 (68) 11 (46) Moderate 3 (13) 6 (26) 5 (23) 8 (33) Severe 0 1 (4) 0 2 (8) TEAEs in ≥10% among all alixorexton-treated patients Pollakiuria 1 (4) 15 (65) 11 (50) 12 (50) Insomnia 0 4 (17) 7 (32) 8 (33) Salivary Hypersecretion 0 5 (22) 5 (23) 7 (29) Micturition Urgency 1 (4) 2 (9) 4 (18) 4 (17) Vision Blurred 1 (4) 2 (9) 1 (5) 7 (29) Drug-related TEAEsa,b 6 (26) 18 (78) 17 (77) 19 (79) Serious TEAEs 0 0 0 0 TEAEs leading to study drug discontinuation 0 0 0 1 (4)
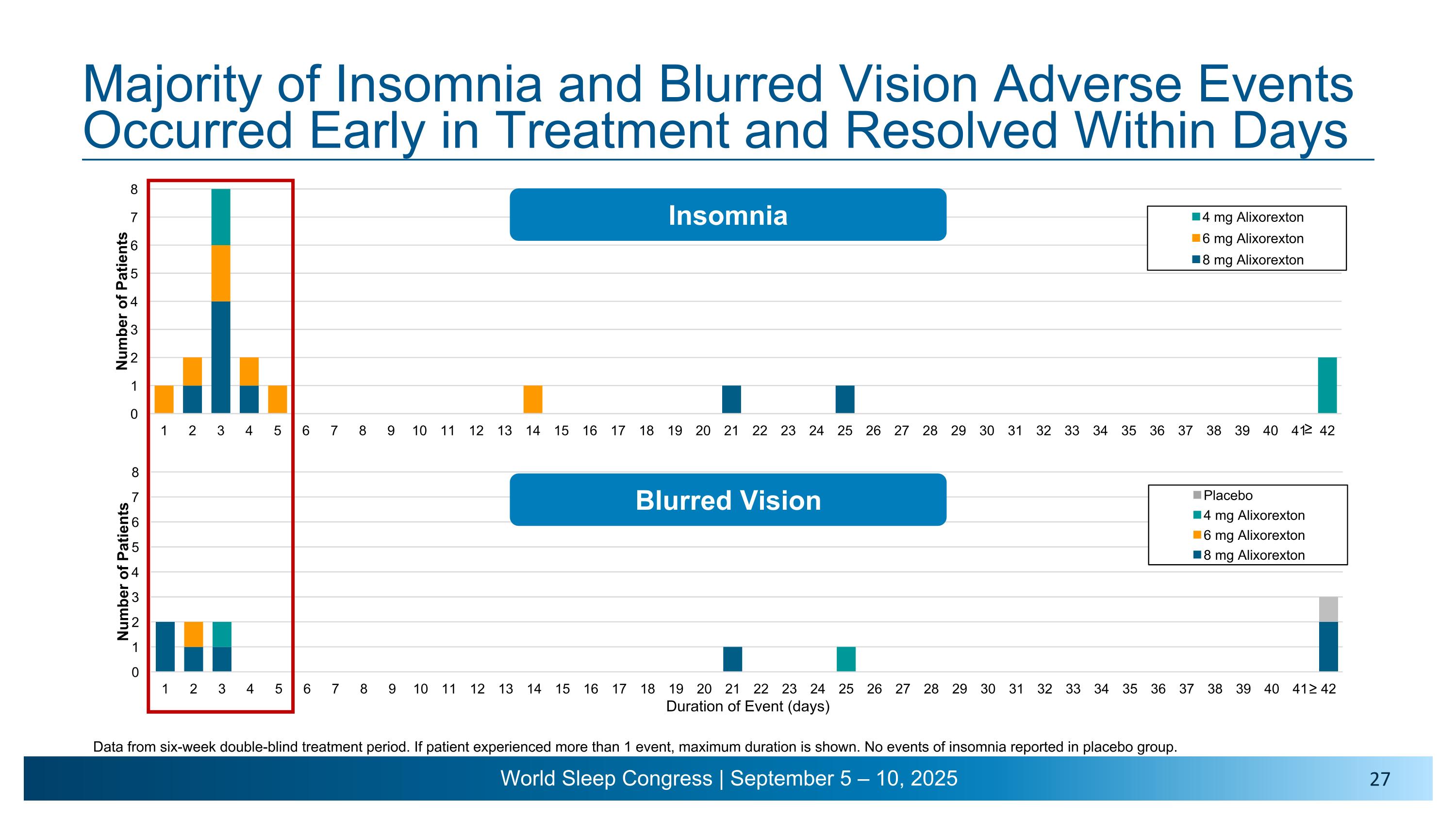
Majority of Insomnia and Blurred Vision Adverse Events Occurred Early in Treatment and Resolved Within Days ≥ Insomnia Blurred Vision ≥ Data from six-week double-blind treatment period. If patient experienced more than 1 event, maximum duration is shown. No events of insomnia reported in placebo group.

Alixorexton was Generally Well Tolerated Over 6 �Weeks of Treatment Most TEAEs were mild to moderate in severity There were no serious TEAEs reported Most commonly reported TEAEs occurred within the 1st week of dosing Most insomnia events resolved within 1 week Most urinary events (pollakiuria and micturition urgency) were generally persistent Most vision blurred events were mild, transient or intermittent, and resolved within 3 days No clinically meaningful changes in patients treated with alixorexton were noted in heart rate, blood pressure, liver function, or visual exams TEAE = treatment-emergent adverse event.
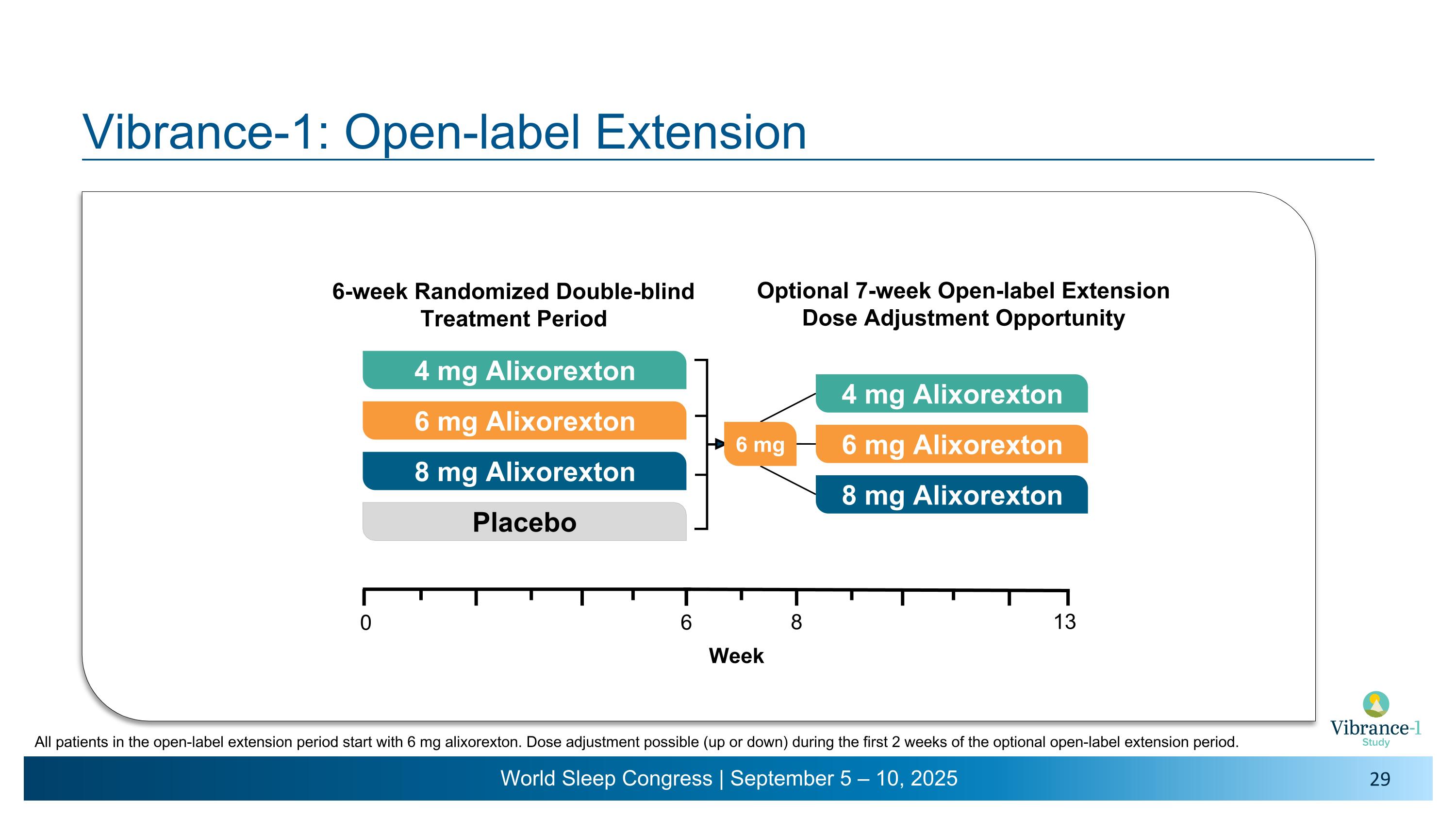
Vibrance-1: Open-label Extension 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo 6-week Randomized Double-blind Treatment Period 6 0 Week 6 mg 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton 8 13 Optional 7-week Open-label Extension�Dose Adjustment Opportunity All patients in the open-label extension period start with 6 mg alixorexton. Dose adjustment possible (up or down) during the first 2 weeks of the optional open-label extension period.
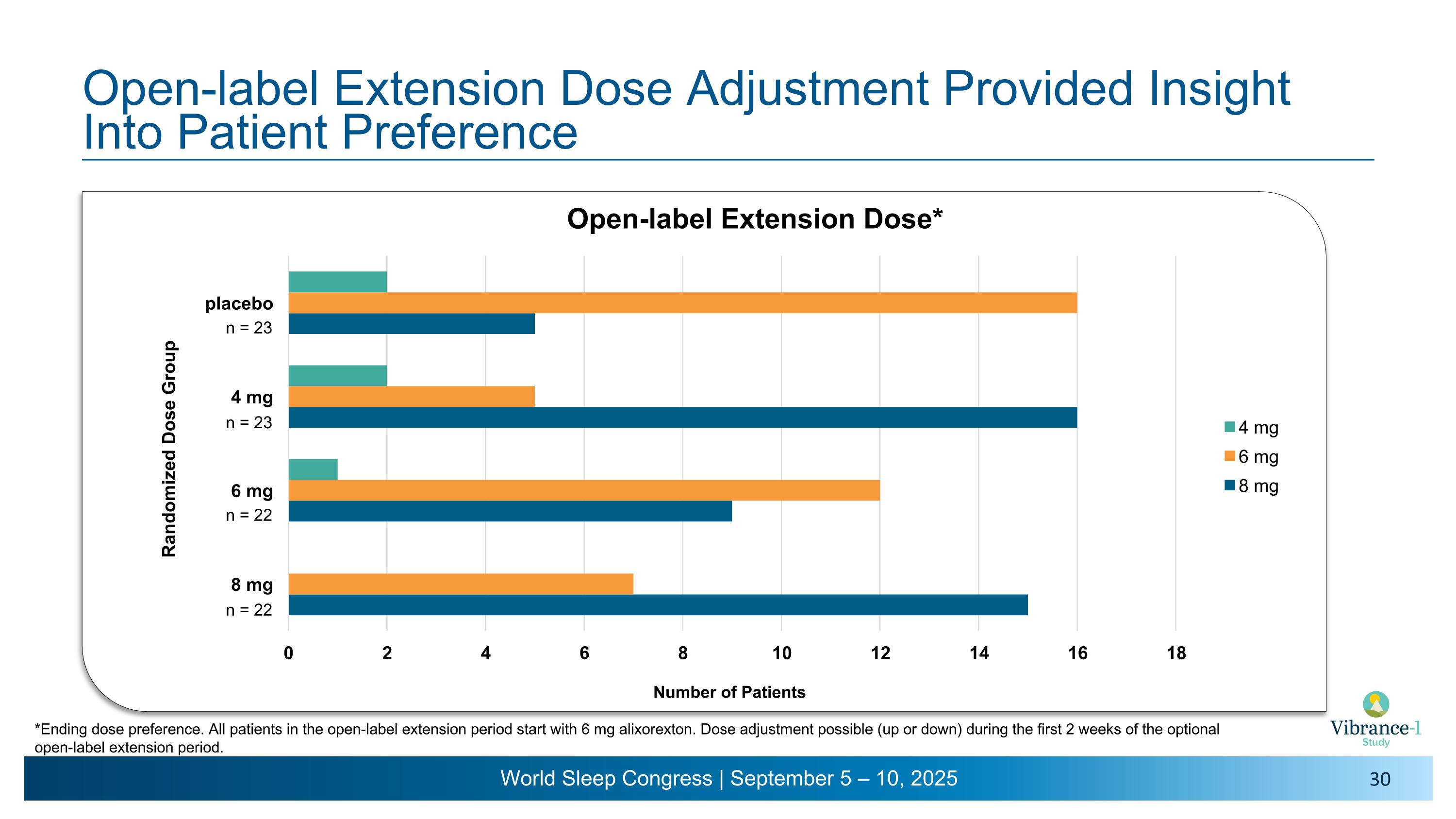
Number of Patients Randomized Dose Group n = 23 n = 23 n = 22 n = 22 Open-label Extension Dose Adjustment Provided Insight Into Patient Preference *Ending dose preference. All patients in the open-label extension period start with 6 mg alixorexton. Dose adjustment possible (up or down) during the first 2 weeks of the optional open-label extension period.

Vibrance-1 Open-label Extension: Safety Summary During the 7-week open-label extension (N=90): Alixorexton was generally well tolerated Most TEAEs were mild to moderate in severity There were no serious TEAEs reported For TEAEs most commonly reported in the double-blind period*, in the open-label extension: Onset of events was primarily associated with treatment initiation (patients who received their first exposure to alixorexton in the open-label extension) Onset of new events was low in patients who had prior alixorexton exposure At the 8 mg dose (n=46 in open-label extension), onset of new events was minimal; �no new events of pollakiuria, insomnia, salivary hypersecretion or blurred vision were reported *Pollakiuria, insomnia, micturition urgency, salivary hypersecretion or blurred vision TEAE = treatment-emergent adverse event.

Vibrance-1 Lead Investigator Director of the Narcolepsy Center at the IRCCS of the Neurological Sciences of Bologna�Professor of Childhood Neuropsychiatry, University of Modena and Reggio Emilia Professor Giuseppe Plazzi, M.D. Ph.D.

= Conclusions Once-daily alixorexton: Demonstrated statistically significant, clinically meaningful improvements on the MWT and ESS at Week 6 compared with placebo, achieving a normative wakefulness profile at all doses tested Improvements on the ESS were observed as early as Week 2 and sustained through Week 13a Reduced WCR at all doses; achieved statistical significance at the 6 mg dose. Many patients had no cataplexy during Week 6 at all doses tested Was generally well tolerated, with most TEAEs mild to moderate in severity and no serious TEAEs reported No clinically meaningful changes in heart rate, blood pressure, liver function, or visual exams Results from Vibrance-1 will inform dose selection for a �planned global phase 3 study in patients with NT1 aBased on data snapshot on 1 July 2025, which included 59 patients who completed the Week 13 visit. ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NT1 = narcolepsy type 1; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

www.alkermes.com

Table of Contents Important Information About This Document This document includes scientific information about alixorexton (formerly referred to as ALKS 2680) that is intended for investors and should be read in conjunction with the press release issued, and investor presentation displayed, by the Company on September 8, 2025. Alixorexton is investigational and has not been approved by the FDA or any other health authority, and its safety and efficacy have not been established. Note Regarding Forward-Looking Statements Certain statements set forth in this document may constitute “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning the potential therapeutic and commercial value of alixorexton (formerly referred to as ALKS 2680) and the company’s expectations regarding the alixorexton development program. Such forward-looking statements are inherently uncertain and, although the company believes that such statements are based on reasonable assumptions within the bounds of its knowledge of its business and operations, these statements are neither promises nor guarantees and they are necessarily subject to a high degree of uncertainty and risk. Actual performance and results may differ materially from those expressed or implied in the forward-looking statements due to various risks and uncertainties. These risks and uncertainties include, among others: whether initial clinical results for alixorexton will be predictive of results of future stages of ongoing clinical studies, future clinical studies or real-world results; whether ongoing or future clinical studies for alixorexton will be initiated or completed on expected timelines or at all; whether alixorexton could be shown to be ineffective or unsafe; potential changes in the cost, scope and duration of the alixorexton development program; and those risks and uncertainties described under the heading “Risk Factors” in the company’s Annual Report on Form 10-K for the year ended Dec. 31, 2024 and in subsequent filings made by the company with the U.S. Securities and Exchange Commission (SEC), which are available on the SEC’s website at www.sec.gov. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this document. Except as required by law, the company disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this document. Alkermes Oral presentations presented at world sleep 2025�September 8, 2025 Vibrance-1: A Randomized Phase 2 Study Evaluating Safety and Efficacy of the Orexin 2 Receptor Agonist Alixorexton (ALKS 2680) in Patients with Narcolepsy Type 1 Improvement in the Severity of Narcolepsy Symptoms and Fatigue in Patients with Narcolepsy Type 1 Treated with the Orexin 2 Receptor Agonist Alixorexton (ALKS 2680) Improvement in Patient-reported Cognitive Functioning in Patients with Narcolepsy Type 1 Treated with the Orexin 2 Receptor Agonist Alixorexton (ALKS 2680) Exhibit 99.3

Giuseppe Plazzi,1 Yves Dauvilliers,2 Ronald R. Grunstein,3 Emmanuel Mignot,4 Gert Jan Lammers,5 David T. Plante,6 Erik Buntinx,7 Rafael del Río Villegas,8 Hailu Chen,9 Alexandra Lovett,9 Craig Hopkinson,9 Bhaskar Rege,9 and Marcus Yountz9 Vibrance-1: A Randomized Phase 2 Study Evaluating �Safety and Efficacy of the Orexin 2 Receptor �Agonist Alixorexton (ALKS 2680) in Patients With �Narcolepsy Type 1 1IRCCS Istituto delle Scienze Neurologiche di Bologna, Bologna, Italy; 2University of Montpellier, INSERM Institute for Neurosciences of Montpellier, Montpellier, France; 3Woolcock Institute of Medical Research, Macquarie University, Sydney, Australia; 4Stanford Center for Sleep Sciences and Medicine, Stanford University School of Medicine, Stanford, CA, USA; 5Leiden University Medical Center, Leiden, the Netherlands and Stichting Epilepsie Instellingen Nederland, Sleep-Wake Centre, Heemstede, the Netherlands; 6University of Wisconsin-Madison, School of Medicine and Public Health, Madison, WI, USA; 7ANIMA Research, Alken, Belgium; 8Universidad CEU San Pablo, CEU Universities, Vithas Madrid Hospitals Madrid, Spain; 9Alkermes, Inc., Waltham, MA, USA

Ineligible companies are those whose primary business is producing, marketing, selling, re-selling, or distributing health care products used by, or on patients. ◻︎ No, I HAVE NOT had a financial relationship with an ineligible company in the past 24 months. ◻︎ Yes, I HAVE had a financial relationship with an ineligible company in the past 24 months. Relationship type Name of company Institutional funding Alkermes (R. Grunstein, Y. Dauvilliers); Lilly (R. Grunstein); Takeda (R. Grunstein); Vanda (R. Grunstein) Research funding Avadel (E. Mignot); Alkermes (E. Mignot); Bioprojet (G. Plazzi); Centessa Pharmaceuticals (G. Plazzi); Eisai (E. Mignot); Idorsia (G. Plazzi); Jazz Pharmaceuticals (G. Plazzi, E. Mignot); Orexia Therapeutics (G. Plazzi); Takeda (G. Plazzi, E. Mignot); Vanda (E. Mignot) Employment Alkermes (A. Lovett, B. Rege, C. Hopkinson, H. Chen, M. Yountz) Speaker fees Eisai (R. Grunstein); SomnoMed (R. Grunstein) Advisory Board Aditum Bio LLC (D. Plante); Alkermes (D. Plante): Apnimed (R. Grunstein); Avadel (Y. Dauvilliers); Bioprojet (Y. Dauvilliers); Centessa (D. Plante, Y. Dauvilliers); Harmony Biosciences (D. Plante, Y. Dauvilliers); Idorsia (Bioprojet); Jazz Pharmaceuticals (D. Plante, Y. Dauvilliers); Lilly (R. Grunstein); Takeda (D. Plante, E. Mignot, Y. Dauvilliers); TEVA (D. Plante).

Orexin 2 Receptor Agonism: Opportunity for Addressing Central Disorders of Hypersomnolence 1. Krahn LE, et al. Adv Ther. 2022;39(1):221-243. 2. Grunstein R, et al. Poster at SLEEP 2025 Meeting; June 8-11, 2025; Seattle, WA. IH = idiopathic hypersomnia; NT1 = narcolepsy type 1; NT2 = narcolepsy type 2. Orexin Regulation of Wakefulness Systems1 In a phase 1b study, alixorexton was generally well tolerated and improved wakefulness across a range of doses in patients with NT1, NT2, and IH, informing dose selection for phase 2 studies2 OREXIN Dopamine Acetylcholine Histamine Serotonin Norepinephrine Alixorexton (ALKS 2680) is a highly potent, oral, selective orexin 2 receptor (OX2R) agonist being developed as a once-daily treatment for narcolepsy and idiopathic hypersomnia Alixorexton has achieved the following design objectives: High potency and selectivity for OX2R in the brain Oral, once-daily dosing that mimics the sleep/wake cycle A wide therapeutic index and range of doses

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.
Vibrance-1 Phase 2 Study: 6-Week Double-blind Period Followed by an Open-label Extension Period With Dosing Flexibility Inclusion criteria Exclusion criteria aAll patients in the open-label extension period start with 6 mg alixorexton. Dose adjustment possible (up or down) during the first 2 weeks of the optional open-label extension period. BMI = body mass index; CSF = cerebrospinal fluid; EDS = Excessive Daytime Sleepiness; ICSD-3-TR = International Classification of Sleep Disorders, third edition, text revision; NT1 = narcolepsy type 1. NT1 patients (ICSD-3-TR) with residual EDS and cataplexy Age 18 to ≤70 years BMI ≥18 and ≤40 kg/m2 HLA-DQB1*06:02-positive or hypocretin-1 CSF ≤110 pg/mL Washout from narcolepsy medications ≥14 days Significant comorbid conditions: Sleep disorders/disturbed sleep Cardiovascular disease Psychiatric or substance use disorder Other chronic conditions (e.g., diabetes, hepatic/renal disease) Optional Open-label Extension 7 Weeks Patients receive alixorexton �(adjusted dosinga) Safety Follow-up 2 Weeks ALKS 2680-301�Phase 2/3 �Long-term �Extension Study 1:1:1:1 Oral, once daily 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo Weeks 2 4 6 0 6-Week Randomized Double-Blind Treatment Period
Primary Efficacy and Secondary Endpoints Were Evaluated at the End of the 6-week Double-blind Treatment Period Weekly cataplexy rate �at Week 6a Change in ESS from baseline to Week 6 Primary Endpoint Change in mean sleep latency on the MWT from baseline to Week 6 Key Secondary Endpoints z z TEAEs Vital signs Laboratory parameters Safety (Secondary Endpoint) Oral, once daily 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo 6-Week Randomized Double-Blind Treatment Period 2 4 6 0 Weeks 1:1:1:1 aWeekly cataplexy rate at Week 6 was derived from patients’ cataplexy diaries over Weeks 5 and 6.�BMI = body mass index; CSF = cerebrospinal fluid; EDS = Excessive Daytime Sleepiness; ESS = Epworth Sleepiness Scale; ICSD-3-TR = International Classification of Sleep Disorders, third edition, text revision; MWT = Maintenance of Wakefulness Test; NT1 = narcolepsy type 1; TEAE = treatment-emergent adverse event. Inclusion criteria Exclusion criteria NT1 patients (ICSD-3-TR) with residual EDS and cataplexy Age 18 to ≤70 years BMI ≥18 and ≤40 kg/m2 HLA-DQB1*06:02-positive or hypocretin-1 CSF ≤110 pg/mL Washout from narcolepsy medications ≥14 days Significant comorbid conditions: Sleep disorders/disturbed sleep Cardiovascular disease Psychiatric or substance use disorder Other chronic conditions (e.g., diabetes, hepatic/renal disease)
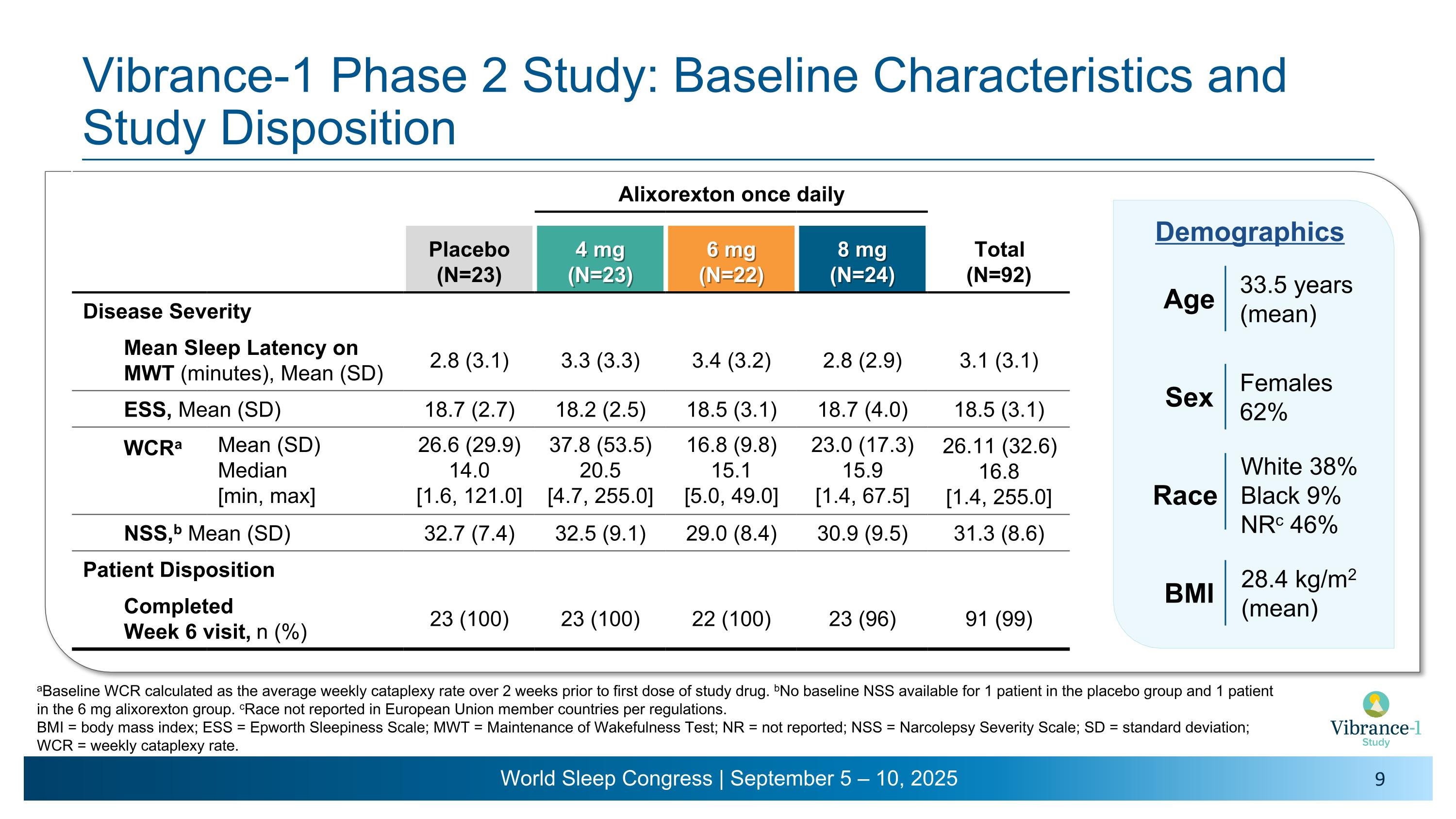
Vibrance-1 Phase 2 Study: Baseline Characteristics and Study Disposition Alixorexton once daily Placebo (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Total (N=92) Disease Severity Mean Sleep Latency on MWT (minutes), Mean (SD) 2.8 (3.1) 3.3 (3.3) 3.4 (3.2) 2.8 (2.9) 3.1 (3.1) ESS, Mean (SD) 18.7 (2.7) 18.2 (2.5) 18.5 (3.1) 18.7 (4.0) 18.5 (3.1) WCRa Mean (SD) Median [min, max] 26.6 (29.9) 14.0 �[1.6, 121.0] 37.8 (53.5) 20.5 �[4.7, 255.0] 16.8 (9.8) 15.1 �[5.0, 49.0] 23.0 (17.3) 15.9 �[1.4, 67.5] 26.11 (32.6) 16.8 [1.4, 255.0] NSS,b Mean (SD) 32.7 (7.4) 32.5 (9.1) 29.0 (8.4) 30.9 (9.5) 31.3 (8.6) Patient Disposition Completed �Week 6 visit, n (%) 23 (100) 23 (100) 22 (100) 23 (96) 91 (99) 33.5 years (mean) Age Females 62% Sex Race White 38% Black 9% NRc 46% 28.4 kg/m2 �(mean) BMI Demographics aBaseline WCR calculated as the average weekly cataplexy rate over 2 weeks prior to first dose of study drug. bNo baseline NSS available for 1 patient in the placebo group and 1 patient in the 6 mg alixorexton group. cRace not reported in European Union member countries per regulations. BMI = body mass index; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NR = not reported; NSS = Narcolepsy Severity Scale; SD = standard deviation; WCR = weekly cataplexy rate.
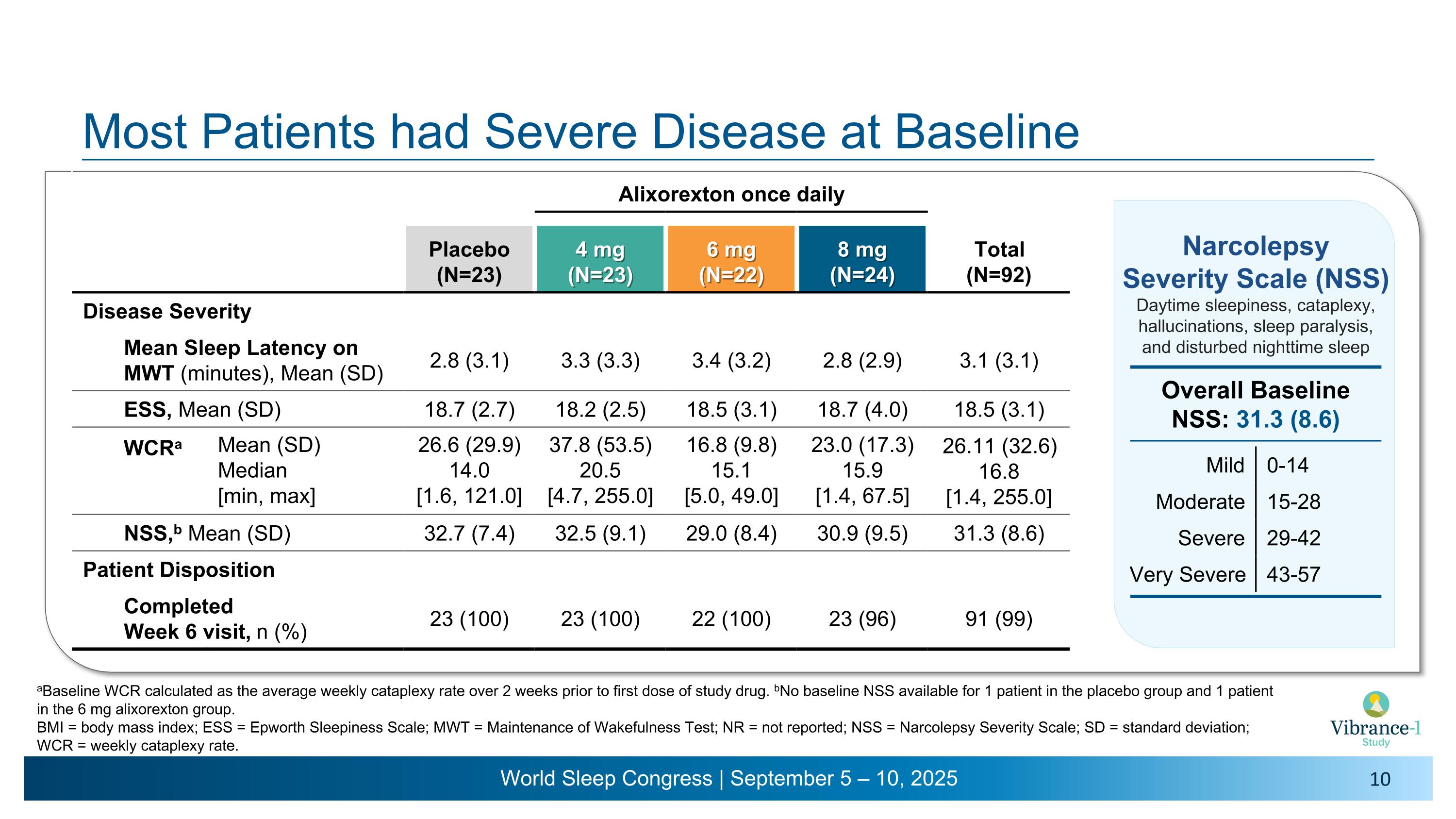
Most Patients had Severe Disease at Baseline Narcolepsy �Severity Scale (NSS) Daytime sleepiness, cataplexy, hallucinations, sleep paralysis, �and disturbed nighttime sleep Mild 0-14 Moderate 15-28 Severe 29-42 Very Severe 43-57 Overall Baseline �NSS: 31.3 (8.6) Alixorexton once daily Placebo (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Total (N=92) Disease Severity Mean Sleep Latency on MWT (minutes), Mean (SD) 2.8 (3.1) 3.3 (3.3) 3.4 (3.2) 2.8 (2.9) 3.1 (3.1) ESS, Mean (SD) 18.7 (2.7) 18.2 (2.5) 18.5 (3.1) 18.7 (4.0) 18.5 (3.1) WCRa Mean (SD) Median [min, max] 26.6 (29.9) 14.0 �[1.6, 121.0] 37.8 (53.5) 20.5 �[4.7, 255.0] 16.8 (9.8) 15.1 �[5.0, 49.0] 23.0 (17.3) 15.9 �[1.4, 67.5] 26.11 (32.6) 16.8 [1.4, 255.0] NSS,b Mean (SD) 32.7 (7.4) 32.5 (9.1) 29.0 (8.4) 30.9 (9.5) 31.3 (8.6) Patient Disposition Completed �Week 6 visit, n (%) 23 (100) 23 (100) 22 (100) 23 (96) 91 (99) aBaseline WCR calculated as the average weekly cataplexy rate over 2 weeks prior to first dose of study drug. bNo baseline NSS available for 1 patient in the placebo group and 1 patient in the 6 mg alixorexton group. BMI = body mass index; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NR = not reported; NSS = Narcolepsy Severity Scale; SD = standard deviation; WCR = weekly cataplexy rate.
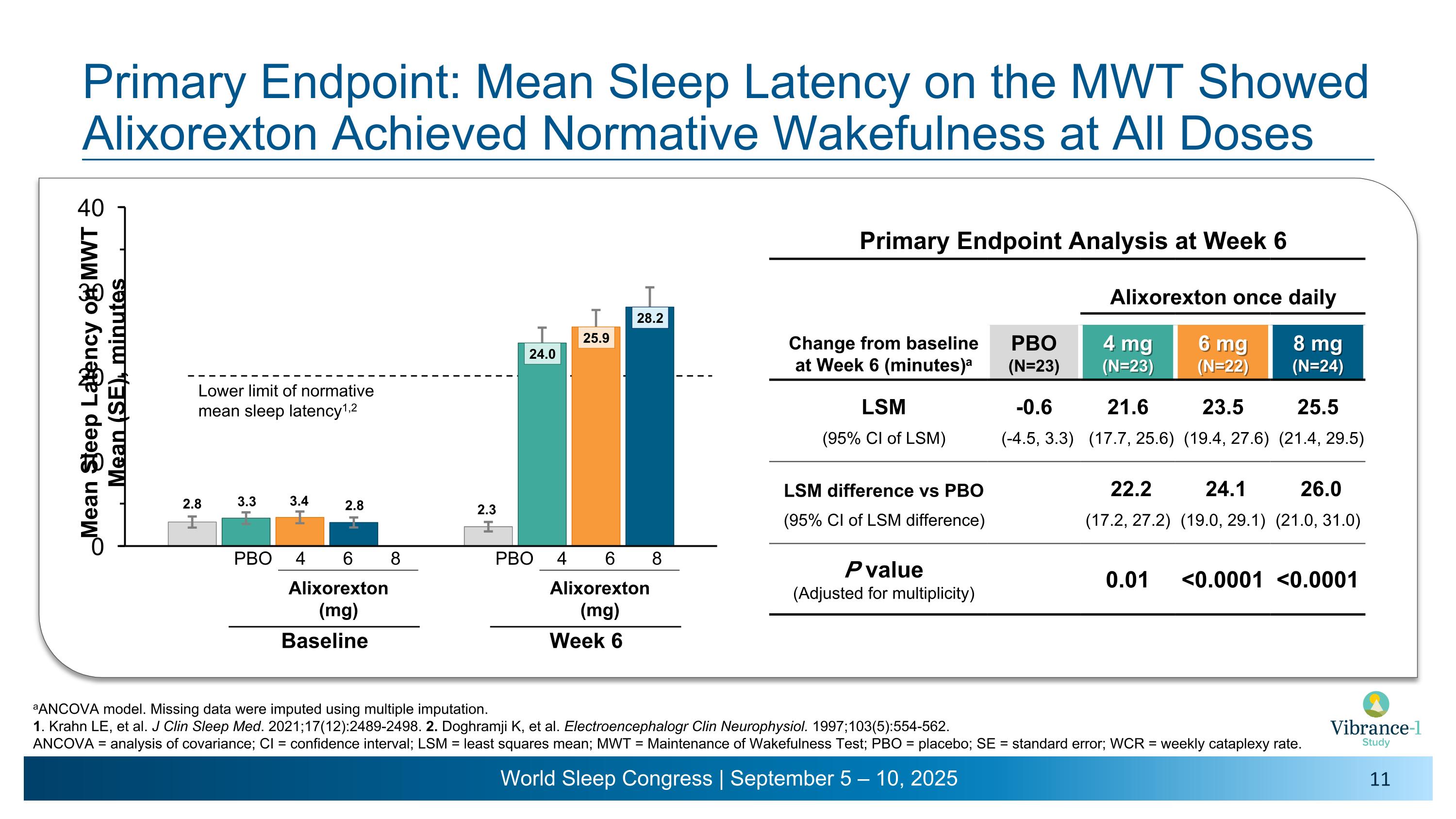
Primary Endpoint: Mean Sleep Latency on the MWT Showed Alixorexton Achieved Normative Wakefulness at All Doses Primary Endpoint Analysis at Week 6 Alixorexton once daily Change from baseline �at Week 6 (minutes)a PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) LSM -0.6 21.6 23.5 25.5 (95% CI of LSM) (-4.5, 3.3) (17.7, 25.6) (19.4, 27.6) (21.4, 29.5) LSM difference vs PBO 22.2 24.1 26.0 (95% CI of LSM difference) (17.2, 27.2) (19.0, 29.1) (21.0, 31.0) P value (Adjusted for multiplicity) 0.01 <0.0001 <0.0001 aANCOVA model. Missing data were imputed using multiple imputation. 1. Krahn LE, et al. J Clin Sleep Med. 2021;17(12):2489-2498. 2. Doghramji K, et al. Electroencephalogr Clin Neurophysiol. 1997;103(5):554-562. ANCOVA = analysis of covariance; CI = confidence interval; LSM = least squares mean; MWT = Maintenance of Wakefulness Test; PBO = placebo; SE = standard error; WCR = weekly cataplexy rate. Lower limit of normative mean sleep latency1,2 4 6 8 PBO Alixorexton �(mg) Baseline 4 6 8 PBO Alixorexton �(mg) Week 6
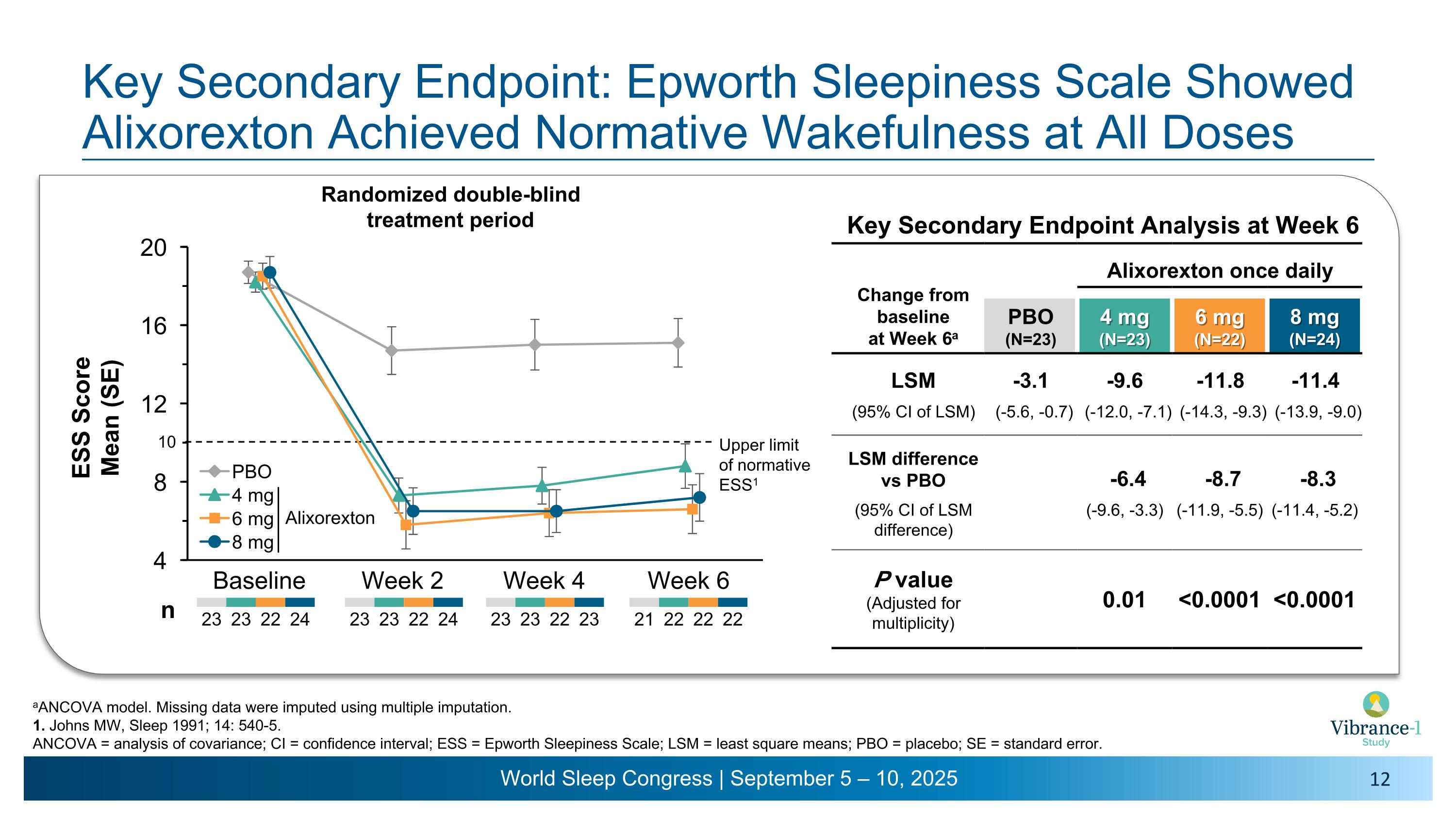
Key Secondary Endpoint: Epworth Sleepiness Scale Showed Alixorexton Achieved Normative Wakefulness at All Doses aANCOVA model. Missing data were imputed using multiple imputation. 1. Johns MW, Sleep 1991; 14: 540-5. ANCOVA = analysis of covariance; CI = confidence interval; ESS = Epworth Sleepiness Scale; LSM = least square means; PBO = placebo; SE = standard error. Baseline Week 2 Week 4 Week 6 23 23 22 24 23 23 22 23 23 23 22 24 n Randomized double-blind �treatment period Alixorexton 21 22 22 22 Key Secondary Endpoint Analysis at Week 6 Change from �baseline �at Week 6a Alixorexton once daily PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) LSM -3.1 -9.6 -11.8 -11.4 (95% CI of LSM) (-5.6, -0.7) (-12.0, -7.1) (-14.3, -9.3) (-13.9, -9.0) LSM difference vs PBO -6.4 -8.7 -8.3 (95% CI of LSM difference) (-9.6, -3.3) (-11.9, -5.5) (-11.4, -5.2) P value (Adjusted for multiplicity) 0.01 <0.0001 <0.0001 Upper limit �of normative ESS1 10
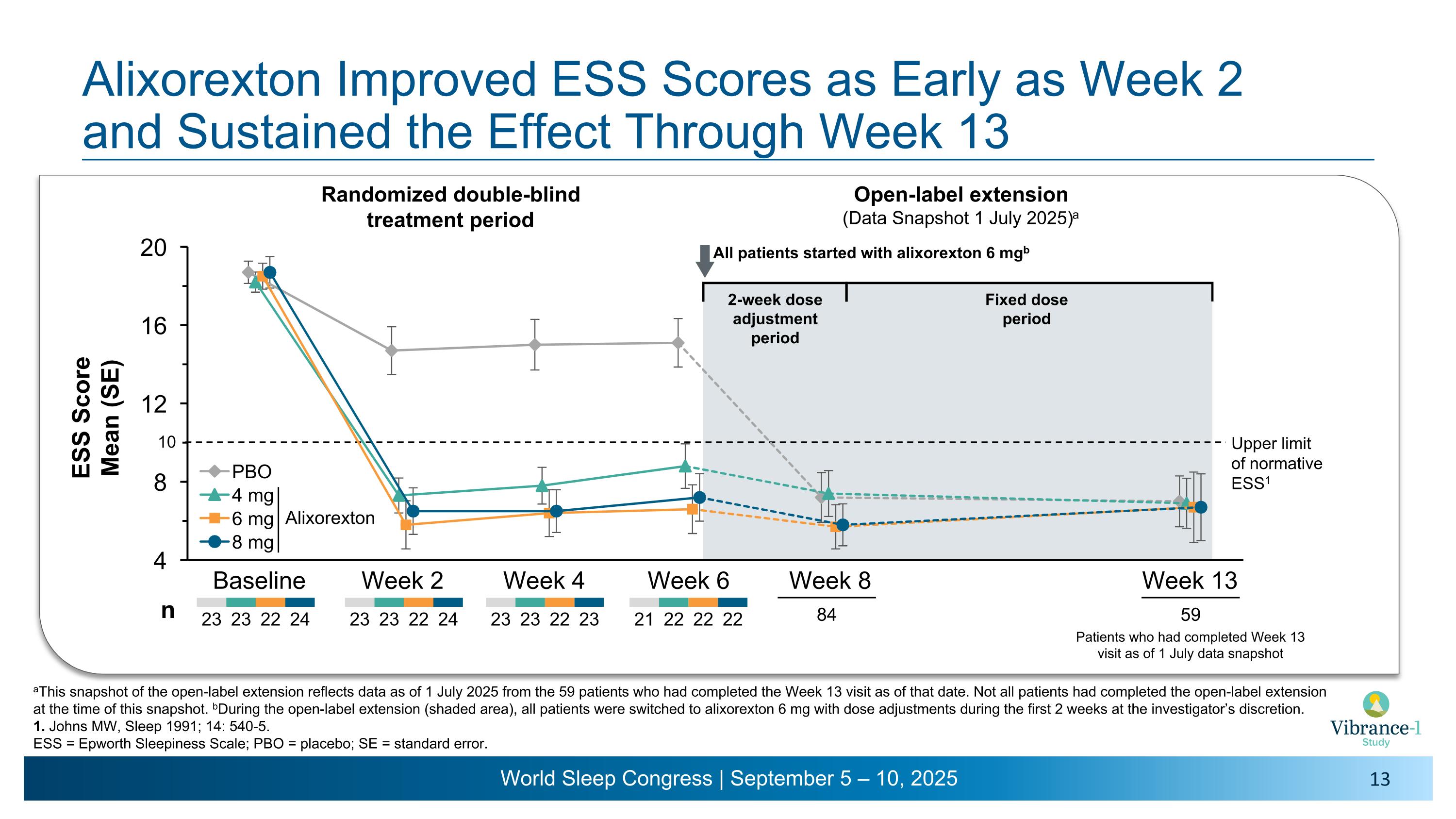
Alixorexton Improved ESS Scores as Early as Week 2 �and Sustained the Effect Through Week 13 aThis snapshot of the open-label extension reflects data as of 1 July 2025 from the 59 patients who had completed the Week 13 visit as of that date. Not all patients had completed the open-label extension at the time of this snapshot. bDuring the open-label extension (shaded area), all patients were switched to alixorexton 6 mg with dose adjustments during the first 2 weeks at the investigator’s discretion. 1. Johns MW, Sleep 1991; 14: 540-5. ESS = Epworth Sleepiness Scale; PBO = placebo; SE = standard error. Baseline Week 2 Week 4 Week 6 Week 8 Upper limit �of normative ESS1 Week 13 23 23 22 24 23 23 22 23 23 23 22 24 84 All patients started with alixorexton 6 mgb n Open-label extension (Data Snapshot 1 July 2025)a Randomized double-blind �treatment period 21 22 22 22 Patients who had completed Week 13 visit as of 1 July data snapshot 59 2-week dose �adjustment period Fixed dose period 10 Alixorexton
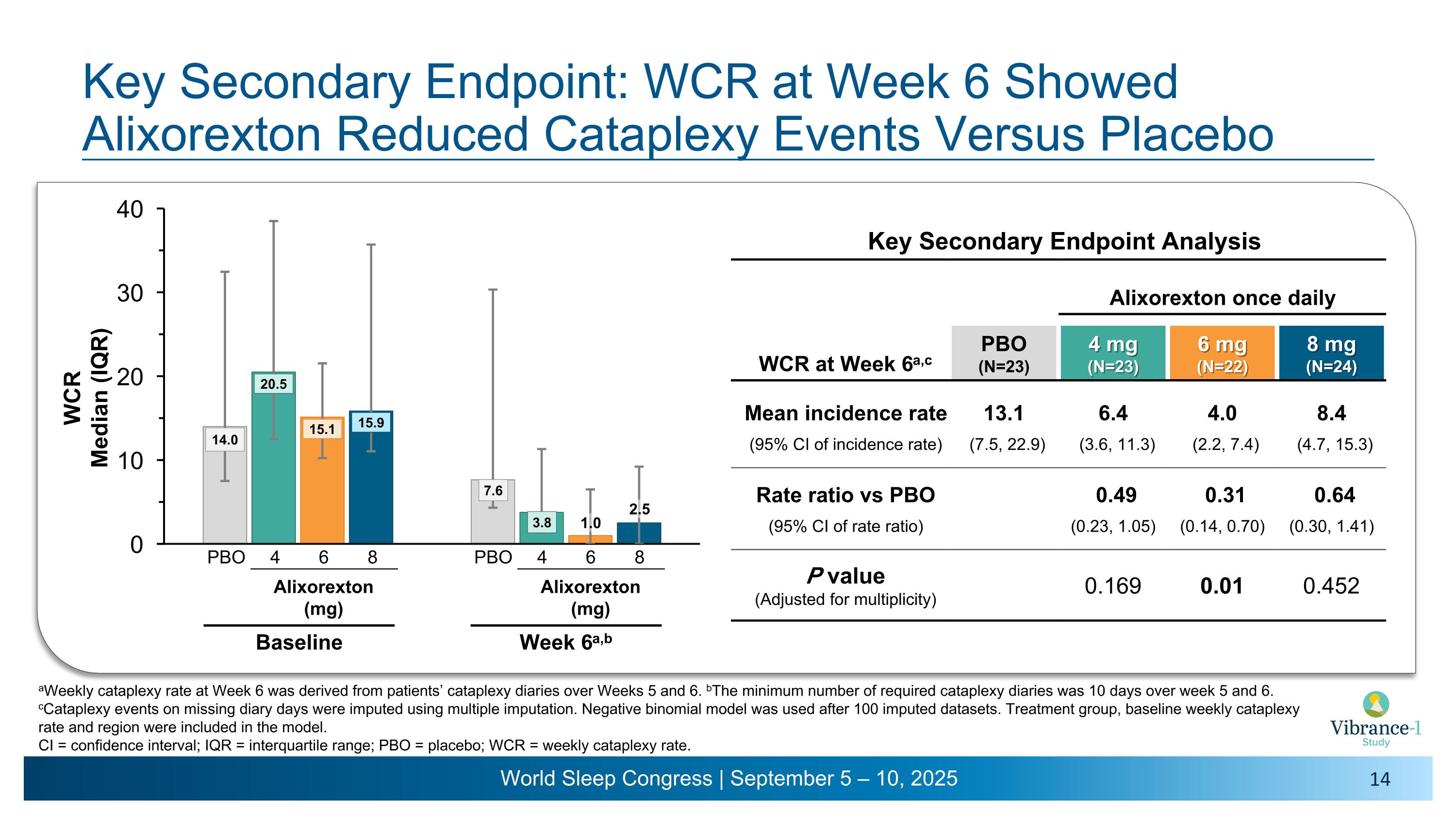
Key Secondary Endpoint: WCR at Week 6 Showed �Alixorexton Reduced Cataplexy Events Versus Placebo Key Secondary Endpoint Analysis Alixorexton once daily WCR at Week 6a,c PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) Mean incidence rate 13.1 6.4 4.0 8.4 (95% CI of incidence rate) (7.5, 22.9) (3.6, 11.3) (2.2, 7.4) (4.7, 15.3) Rate ratio vs PBO 0.49 0.31 0.64 (95% CI of rate ratio) (0.23, 1.05) (0.14, 0.70) (0.30, 1.41) P value (Adjusted for multiplicity) 0.169 0.01 0.452 aWeekly cataplexy rate at Week 6 was derived from patients’ cataplexy diaries over Weeks 5 and 6. bThe minimum number of required cataplexy diaries was 10 days over week 5 and 6. cCataplexy events on missing diary days were imputed using multiple imputation. Negative binomial model was used after 100 imputed datasets. Treatment group, baseline weekly cataplexy rate and region were included in the model. �CI = confidence interval; IQR = interquartile range; PBO = placebo; WCR = weekly cataplexy rate. 1.0 2.5 4 6 8 PBO Alixorexton �(mg) Baseline 4 6 8 PBO Alixorexton �(mg) Week 6a,b
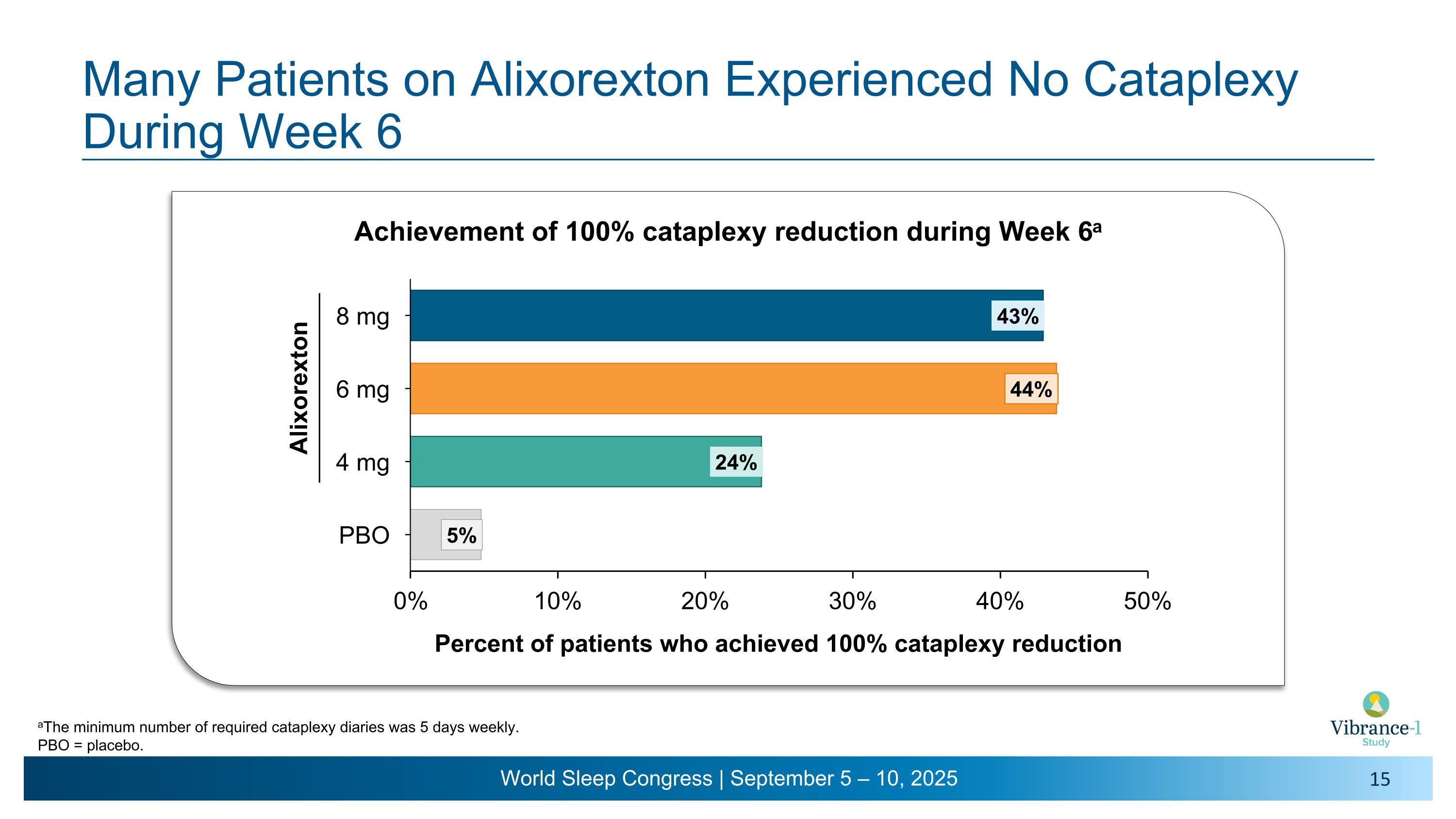
Many Patients on Alixorexton Experienced No Cataplexy During Week 6 = Alixorexton Achievement of 100% cataplexy reduction during Week 6a aThe minimum number of required cataplexy diaries was 5 days weekly. PBO = placebo.
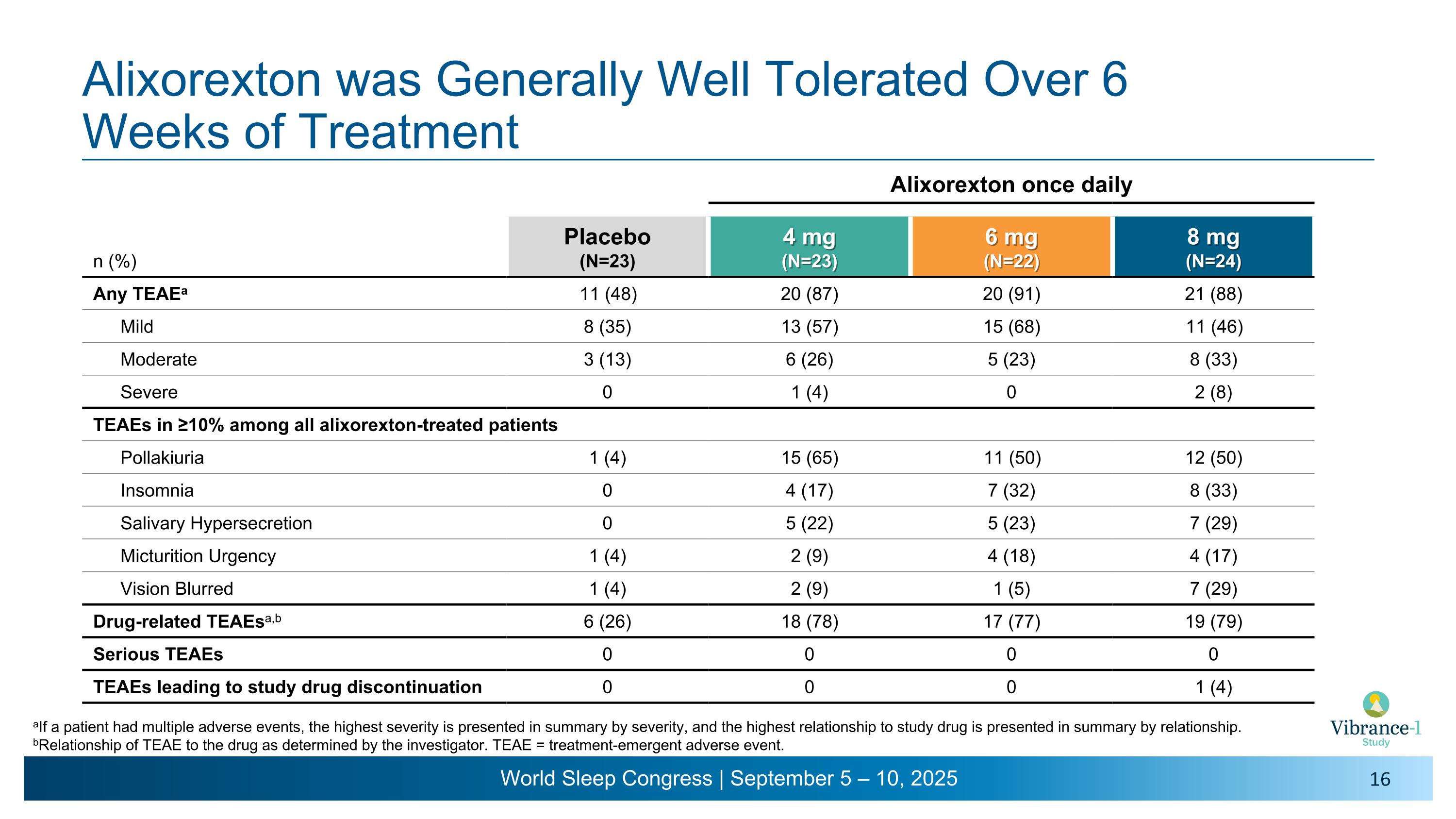
Alixorexton was Generally Well Tolerated Over 6 �Weeks of Treatment aIf a patient had multiple adverse events, the highest severity is presented in summary by severity, and the highest relationship to study drug is presented in summary by relationship. bRelationship of TEAE to the drug as determined by the investigator. TEAE = treatment-emergent adverse event. Alixorexton once daily n (%) Placebo (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Any TEAEa 11 (48) 20 (87) 20 (91) 21 (88) Mild 8 (35) 13 (57) 15 (68) 11 (46) Moderate 3 (13) 6 (26) 5 (23) 8 (33) Severe 0 1 (4) 0 2 (8) TEAEs in ≥10% among all alixorexton-treated patients Pollakiuria 1 (4) 15 (65) 11 (50) 12 (50) Insomnia 0 4 (17) 7 (32) 8 (33) Salivary Hypersecretion 0 5 (22) 5 (23) 7 (29) Micturition Urgency 1 (4) 2 (9) 4 (18) 4 (17) Vision Blurred 1 (4) 2 (9) 1 (5) 7 (29) Drug-related TEAEsa,b 6 (26) 18 (78) 17 (77) 19 (79) Serious TEAEs 0 0 0 0 TEAEs leading to study drug discontinuation 0 0 0 1 (4)

Alixorexton was Generally Well Tolerated Over 6 �Weeks of Treatment Most TEAEs were mild to moderate in severity There were no serious TEAEs reported Most commonly reported TEAEs occurred within the 1st week of dosing Most insomnia events resolved within 1 week Most urinary events (pollakiuria and micturition urgency) were generally persistent Most vision blurred events were mild, transient or intermittent, and resolved within 3 days No clinically meaningful changes in patients treated with alixorexton were noted in heart rate, blood pressure, liver function, or visual exams TEAE = treatment-emergent adverse event.

= Conclusions Once-daily alixorexton: Demonstrated statistically significant, clinically meaningful improvements on the MWT and ESS at Week 6 compared with placebo, achieving a normative wakefulness profile at all doses tested Improvements on the ESS were observed as early as Week 2 and sustained through Week 13a Reduced WCR at all doses; achieved statistical significance at the 6 mg dose. Many patients had no cataplexy during Week 6 at all doses tested Was generally well tolerated, with most TEAEs mild to moderate in severity and no serious TEAEs reported No clinically meaningful changes in heart rate, blood pressure, liver function, or visual exams Results from Vibrance-1 will inform dose selection for a �planned global phase 3 study in patients with NT1 aBased on data snapshot on 1 July 2025, which included 59 patients who completed the Week 13 visit. ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NT1 = narcolepsy type 1; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Demonstrated Clinically Meaningful Improvements on Outcomes Important to Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Acknowledgments The authors would like to thank: The patients Their families and caregivers Vibrance-1 investigators and their staff Funding provided by Alkermes

Yves Dauvilliers,1 Ronald R. Grunstein,2 Emmanuel Mignot,3 Gert Jan Lammers,4 David T. Plante5 Erik Buntinx,6 Rafael del Río Villegas,7 Hailu Chen,8 Craig Hopkinson,8 Bhaskar Rege,8 Marcus Yountz,8 Michael J. Doane,8 and Giuseppe Plazzi9 Improvement in the Severity of Narcolepsy Symptoms and Fatigue in Patients With Narcolepsy Type 1 Treated With the Orexin 2 Receptor Agonist Alixorexton �(ALKS 2680) 1University of Montpellier, INSERM Institute for Neurosciences of Montpellier, Montpellier, France; 2Woolcock Institute of Medical Research, Macquarie University, Sydney, Australia; 3Stanford Center for Sleep Sciences and Medicine, Stanford University School of Medicine, Stanford, CA, USA; 4Leiden University Medical Center, Leiden, the Netherlands and Stichting Epilepsie Instellingen Nederland, Sleep-Wake Centre, Heemstede, the Netherlands; 5University of Wisconsin-Madison, School of Medicine and Public Health, Madison, WI, USA; 6ANIMA Research, Alken, Belgium; 7Universidad CEU San Pablo, CEU Universities, Vithas Madrid Hospitals Madrid, Spain; 8Alkermes, Inc., Waltham, MA, USA; 9IRCCS Istituto delle Scienze Neurologiche di Bologna, Bologna, Italy

Ineligible companies are those whose primary business is producing, marketing, selling, re-selling, or distributing health care products used by, or on patients. ◻︎ No, I HAVE NOT had a financial relationship with an ineligible company in the past 24 months. ◻︎ Yes, I HAVE had a financial relationship with an ineligible company in the past 24 months. Relationship type Name of company Institutional funding Alkermes (R. Grunstein, Y. Dauvilliers); Lilly (R. Grunstein); Takeda (R. Grunstein); Vanda (R. Grunstein) Research funding Avadel (E. Mignot); Alkermes (E. Mignot); Bioprojet (G. Plazzi); Centessa Pharmaceuticals (G. Plazzi); Eisai (E. Mignot); Idorsia (G. Plazzi); Jazz Pharmaceuticals (G. Plazzi, E. Mignot); Orexia Therapeutics (G. Plazzi); Takeda (G. Plazzi, E. Mignot); Vanda (E. Mignot) Employment Alkermes (B. Rege, C. Hopkinson, H. Chen, M. Doane, M. Yountz) Speaker fees Eisai (R. Grunstein); SomnoMed (R. Grunstein) Advisory Board Aditum Bio LLC (D. Plante); Alkermes (D. Plante): Apnimed (R. Grunstein); Avadel (Y. Dauvilliers); Bioprojet (Y. Dauvilliers); Centessa (D. Plante, Y. Dauvilliers); Harmony Biosciences (D. Plante, Y. Dauvilliers); Idorsia (Bioprojet); Jazz Pharmaceuticals (D. Plante, Y. Dauvilliers); Lilly (R. Grunstein); Takeda (D. Plante, E. Mignot, Y. Dauvilliers); TEVA (D. Plante).
Vibrance-1 Examined the Effects of Alixorexton on Overall Disease Severity and Fatigue in Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.
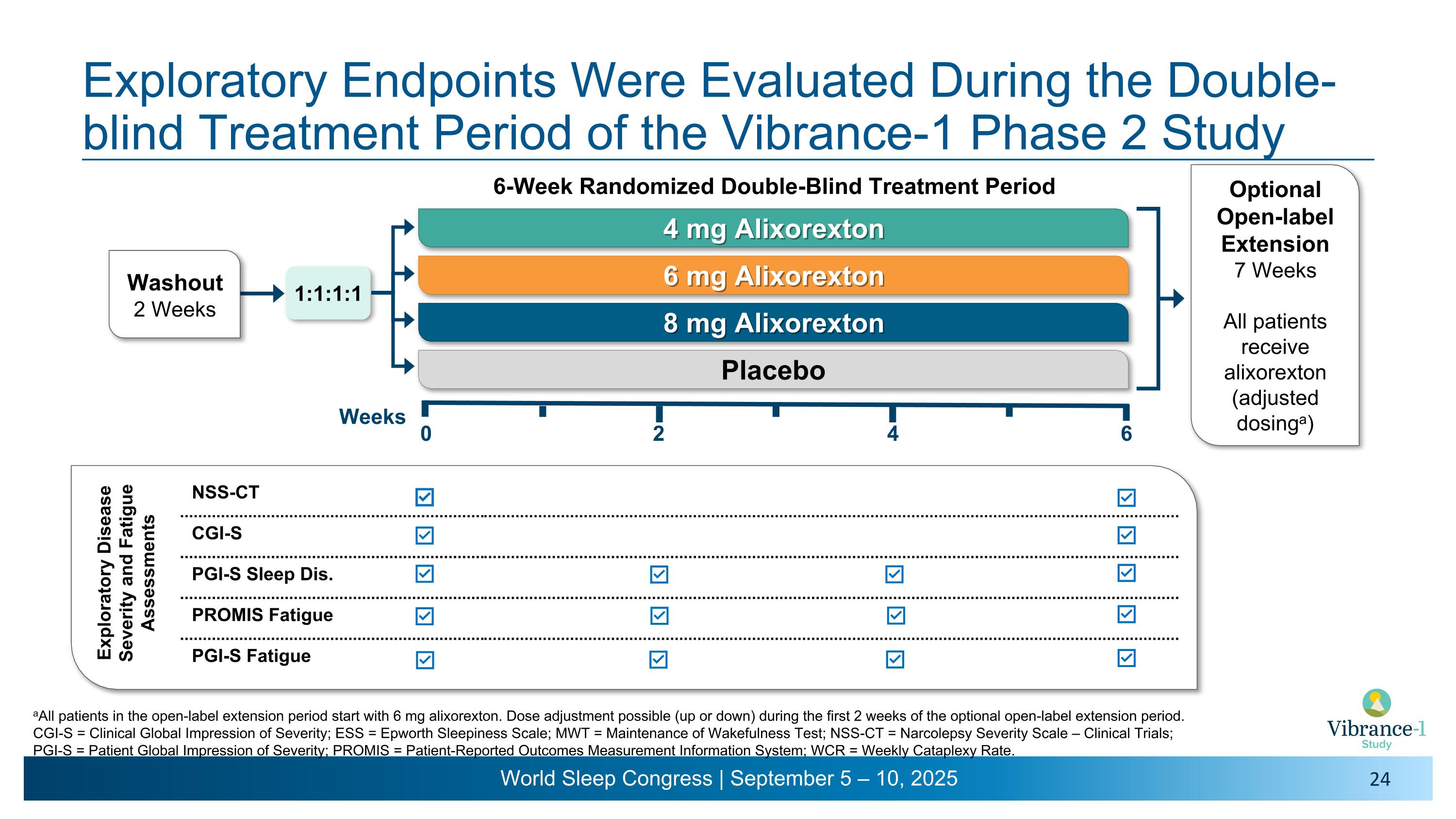
0 Weeks Optional Open-label Extension 7 Weeks All patients receive alixorexton �(adjusted dosinga) Exploratory Endpoints Were Evaluated During the Double-blind Treatment Period of the Vibrance-1 Phase 2 Study 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo 6-Week Randomized Double-Blind Treatment Period 1:1:1:1 2 4 6 NSS-CT CGI-S PGI-S Sleep Dis. PROMIS Fatigue PGI-S Fatigue Washout 2 Weeks aAll patients in the open-label extension period start with 6 mg alixorexton. Dose adjustment possible (up or down) during the first 2 weeks of the optional open-label extension period. CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS-CT = Narcolepsy Severity Scale – Clinical Trials; �PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; WCR = Weekly Cataplexy Rate. Exploratory Disease Severity and Fatigue Assessments
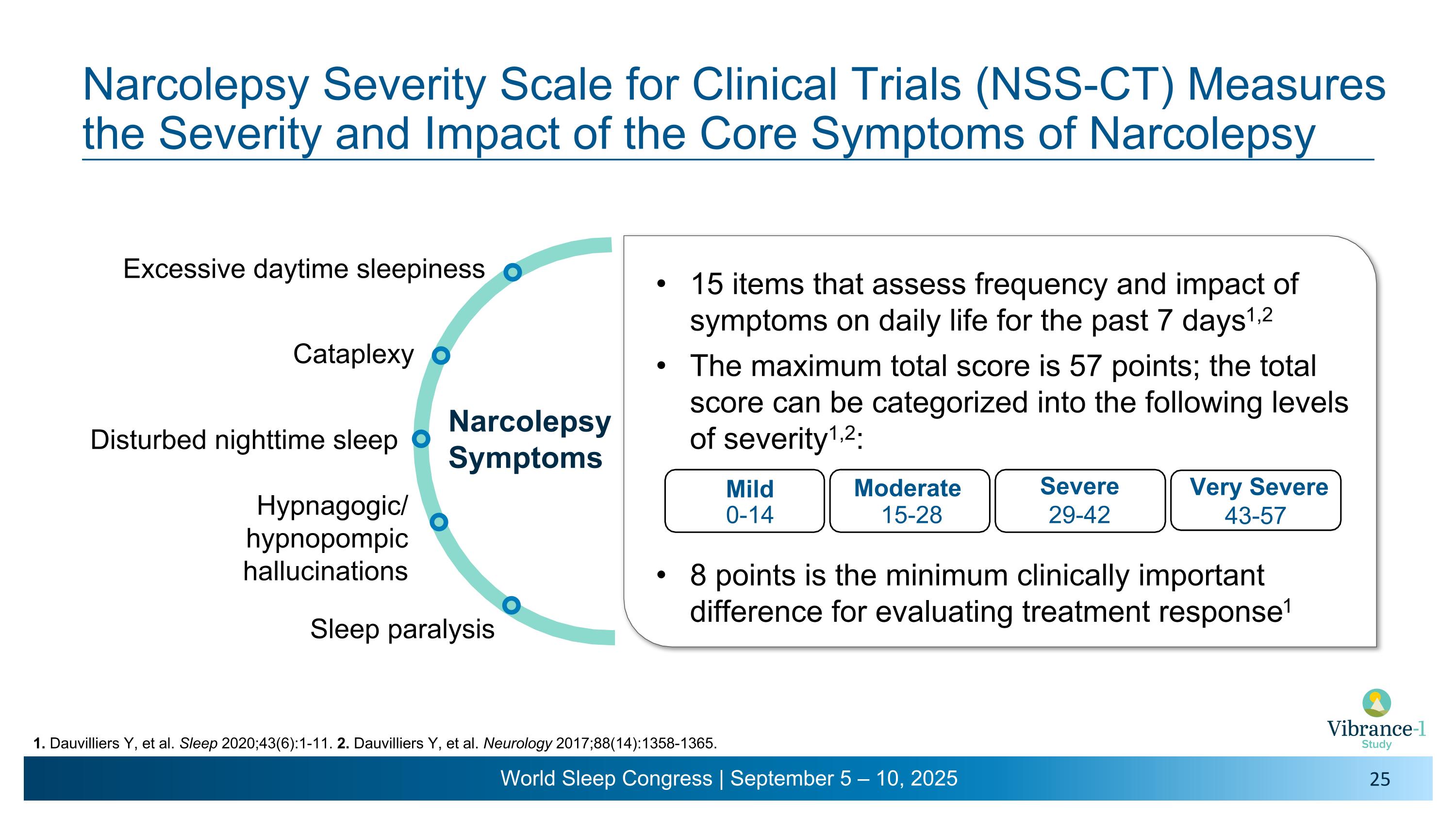
Narcolepsy Symptoms Excessive daytime sleepiness Cataplexy Hypnagogic/�hypnopompic hallucinations Sleep paralysis Narcolepsy Severity Scale for Clinical Trials (NSS-CT) Measures the Severity and Impact of the Core Symptoms of Narcolepsy 1. Dauvilliers Y, et al. Sleep 2020;43(6):1-11. 2. Dauvilliers Y, et al. Neurology 2017;88(14):1358-1365. Disturbed nighttime sleep 15 items that assess frequency and impact of symptoms on daily life for the past 7 days1,2 The maximum total score is 57 points; the total score can be categorized into the following levels of severity1,2: 8 points is the minimum clinically important difference for evaluating treatment response1 Mild Moderate Severe 0-14 15-28 29-42 Very Severe 43-57
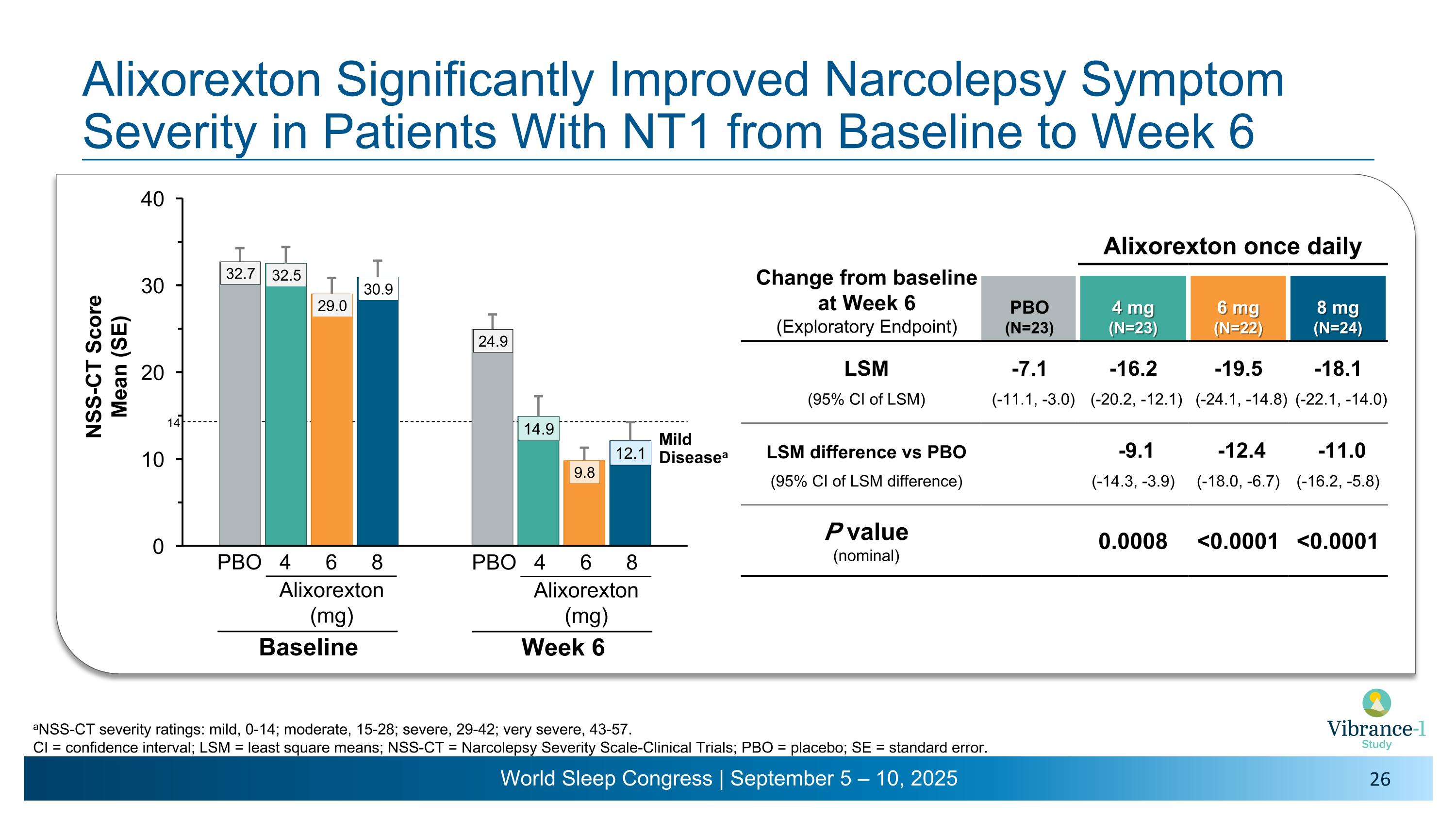
Alixorexton Significantly Improved Narcolepsy Symptom Severity in Patients With NT1 from Baseline to Week 6 aNSS-CT severity ratings: mild, 0-14; moderate, 15-28; severe, 29-42; very severe, 43-57. CI = confidence interval; LSM = least square means; NSS-CT = Narcolepsy Severity Scale-Clinical Trials; PBO = placebo; SE = standard error. Change from baseline �at Week 6 �(Exploratory Endpoint) Alixorexton once daily PBO (N=23) 4 mg�(N=23) 6 mg�(N=22) 8 mg�(N=24) LSM -7.1 -16.2 -19.5 -18.1 (95% CI of LSM) (-11.1, -3.0) (-20.2, -12.1) (-24.1, -14.8) (-22.1, -14.0) LSM difference vs PBO -9.1 -12.4 -11.0 (95% CI of LSM difference) (-14.3, -3.9) (-18.0, -6.7) (-16.2, -5.8) P value (nominal) 0.0008 <0.0001 <0.0001 4 6 8 PBO Alixorexton �(mg) Baseline Mild Diseasea 14 4 6 8 PBO Alixorexton �(mg) Week 6
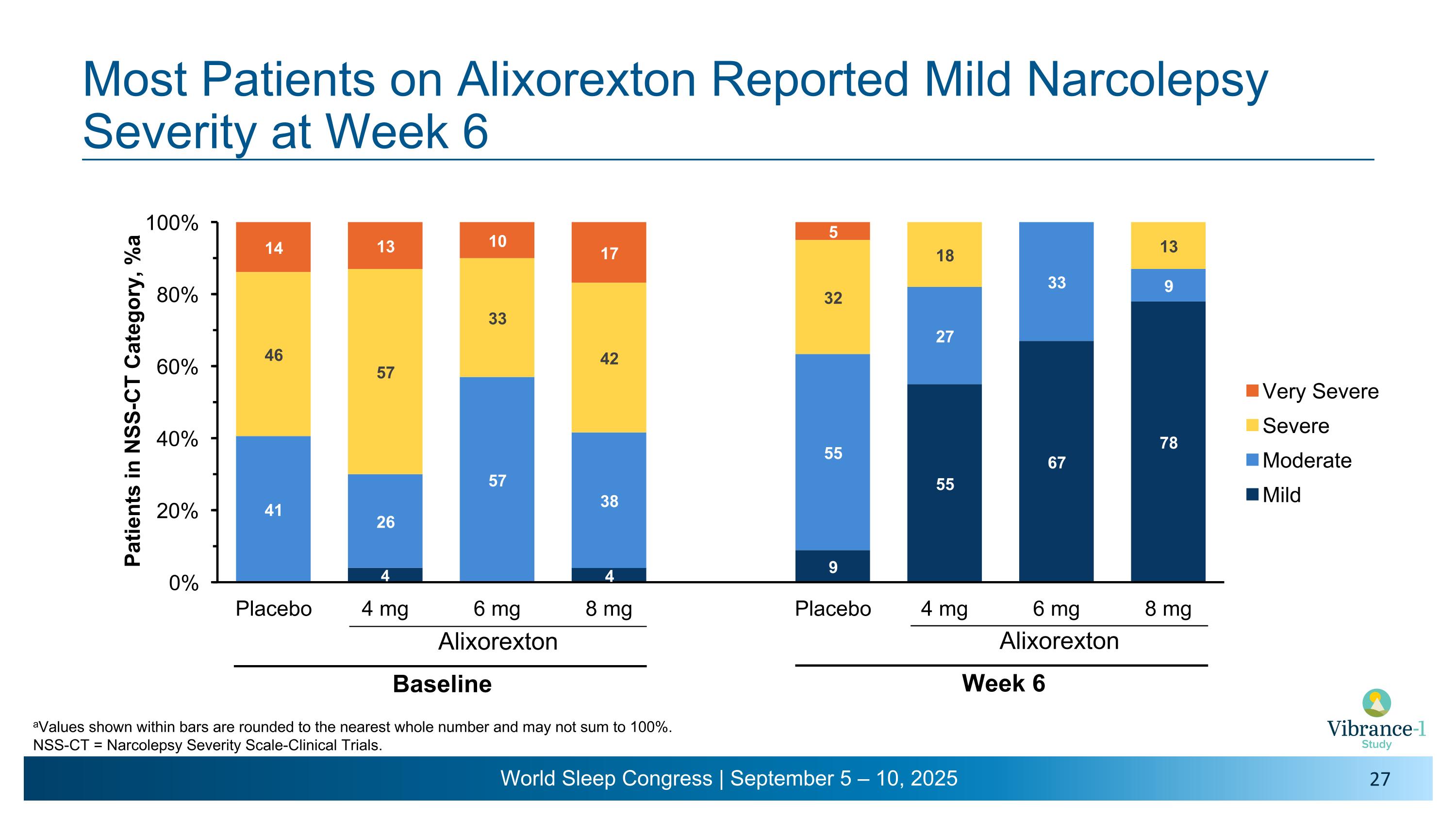
Most Patients on Alixorexton Reported Mild Narcolepsy Severity at Week 6 Alixorexton Baseline aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. NSS-CT = Narcolepsy Severity Scale-Clinical Trials. Alixorexton Week 6
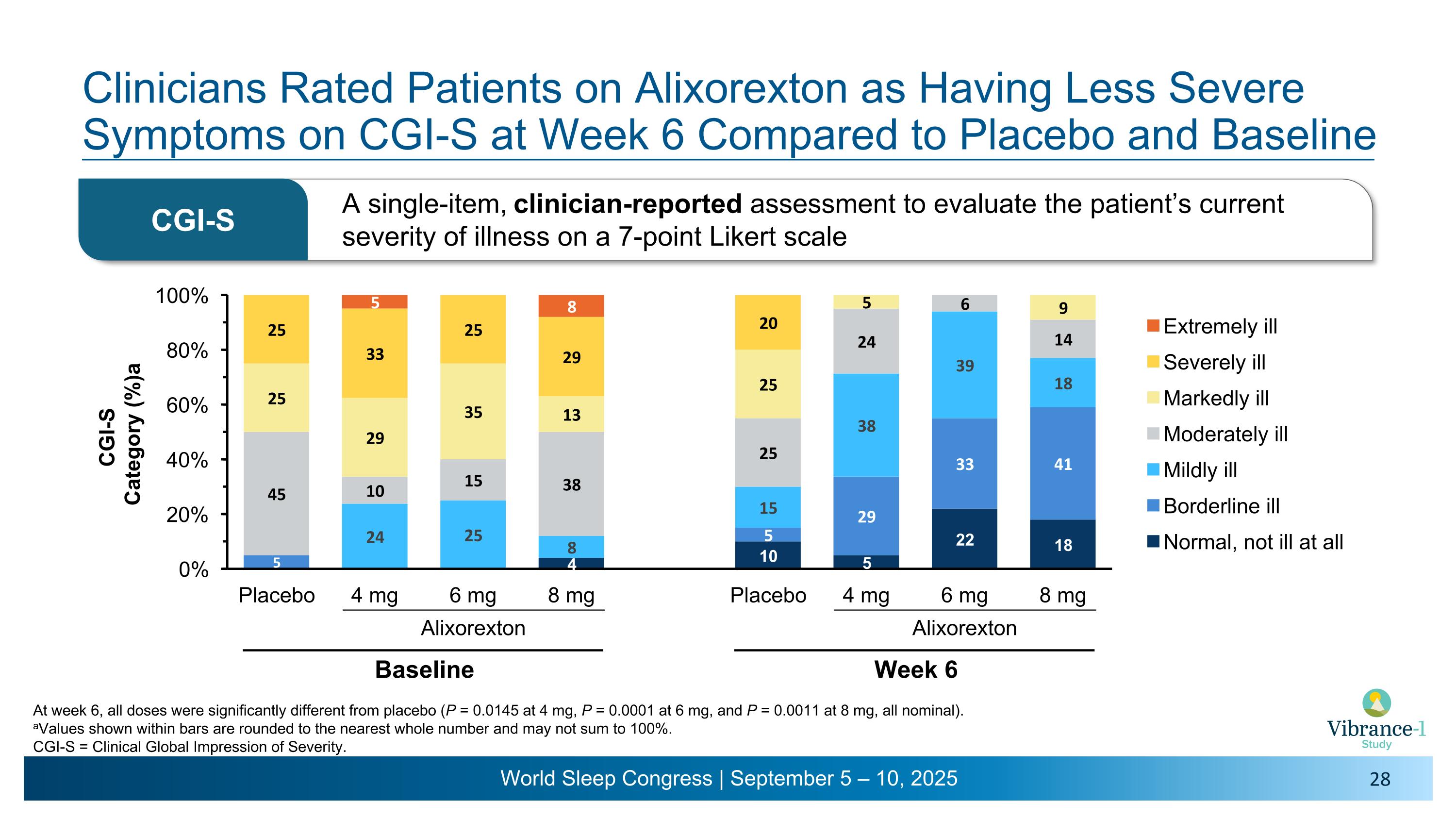
Clinicians Rated Patients on Alixorexton as Having Less Severe Symptoms on CGI-S at Week 6 Compared to Placebo and Baseline Alixorexton Baseline At week 6, all doses were significantly different from placebo (P = 0.0145 at 4 mg, P = 0.0001 at 6 mg, and P = 0.0011 at 8 mg, all nominal). aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. CGI-S = Clinical Global Impression of Severity. A single-item, clinician-reported assessment to evaluate the patient’s current severity of illness on a 7-point Likert scale CGI-S Alixorexton Week 6
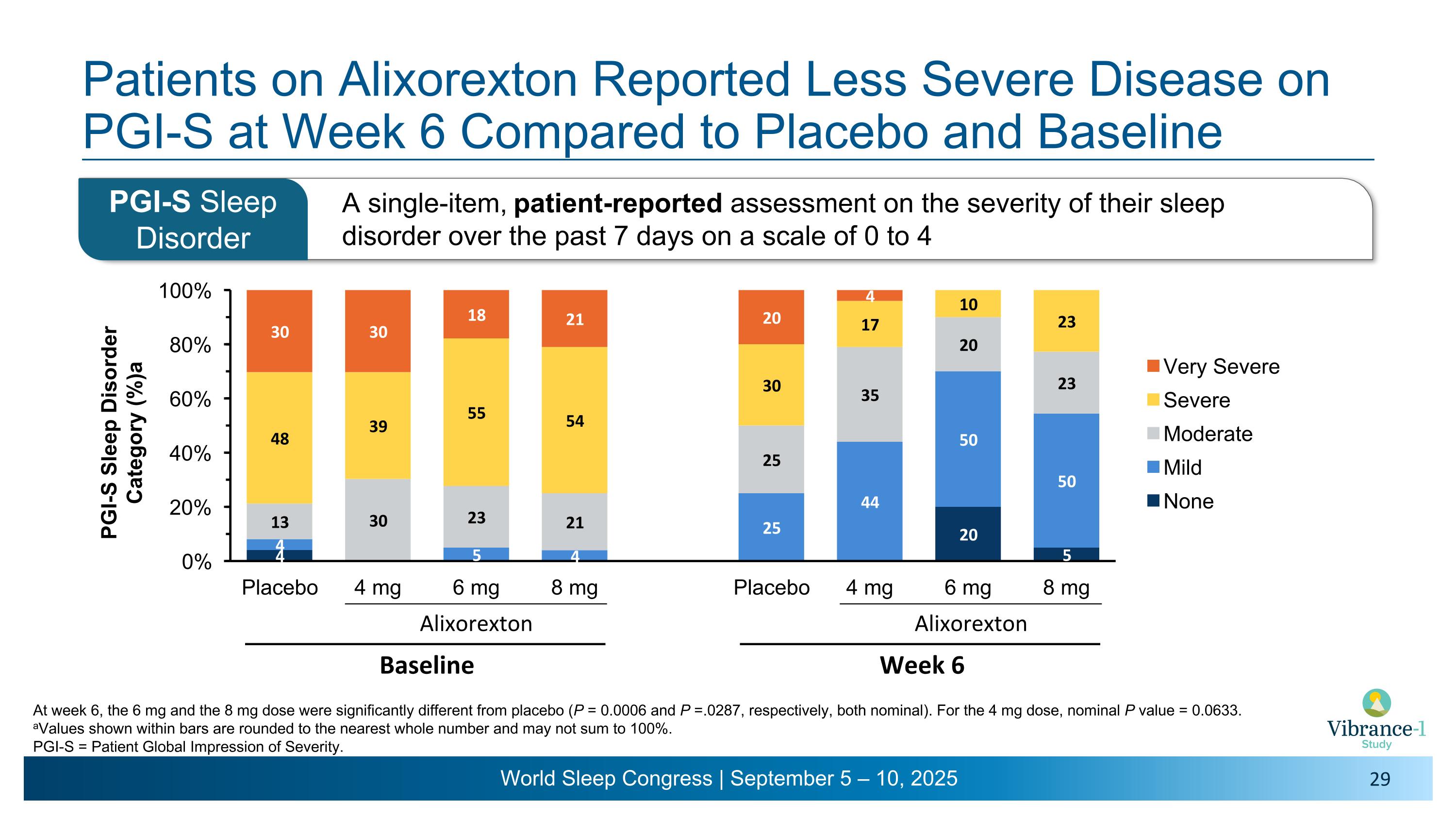
Patients on Alixorexton Reported Less Severe Disease on PGI-S at Week 6 Compared to Placebo and Baseline A single-item, patient-reported assessment on the severity of their sleep disorder over the past 7 days on a scale of 0 to 4 PGI-S Sleep Disorder At week 6, the 6 mg and the 8 mg dose were significantly different from placebo (P = 0.0006 and P =.0287, respectively, both nominal). For the 4 mg dose, nominal P value = 0.0633. aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. PGI-S = Patient Global Impression of Severity. Alixorexton Baseline Alixorexton Week 6

Fatigue Is a Persistent, Severe, and Debilitating Unmet Need That Impacts the Lives of Patients With Narcolepsy Most narcolepsy patients experience fatigue, which often persists even with treatment1-3 Fatigue negatively affects patients’ mental health, functional outcomes and overall quality of life4 PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System.�1. AASM 2023. International Classification of Sleep Disorders – Third Edition: Text Revision, Darien, IL. 2. Maski K, et al. J Clin Sleep Med 2017;13(3):419-425. 3. Doane M, et al. Poster presented at Psych Congress 2023. 4. Droogleever F, et al. Sleep, 21(2), 163-169. PROMIS-Fatigue 6a Short Form A 6-item questionnaire scored on a 5-point Likert scale assessing the severity of fatigue in the past 7 days Items are scored and transformed to T-scores, ranging from 33.4 to 76.8 Scores less than 55 are considered normal, while scores ≥70 are considered severe A single item assessing patient-reported severity of fatigue over the past 7 days with scale responses of none, mild, moderate, severe, or very severe PGI-S Fatigue Commonly Used and Established Patient-reported Measures Specific to Fatigue
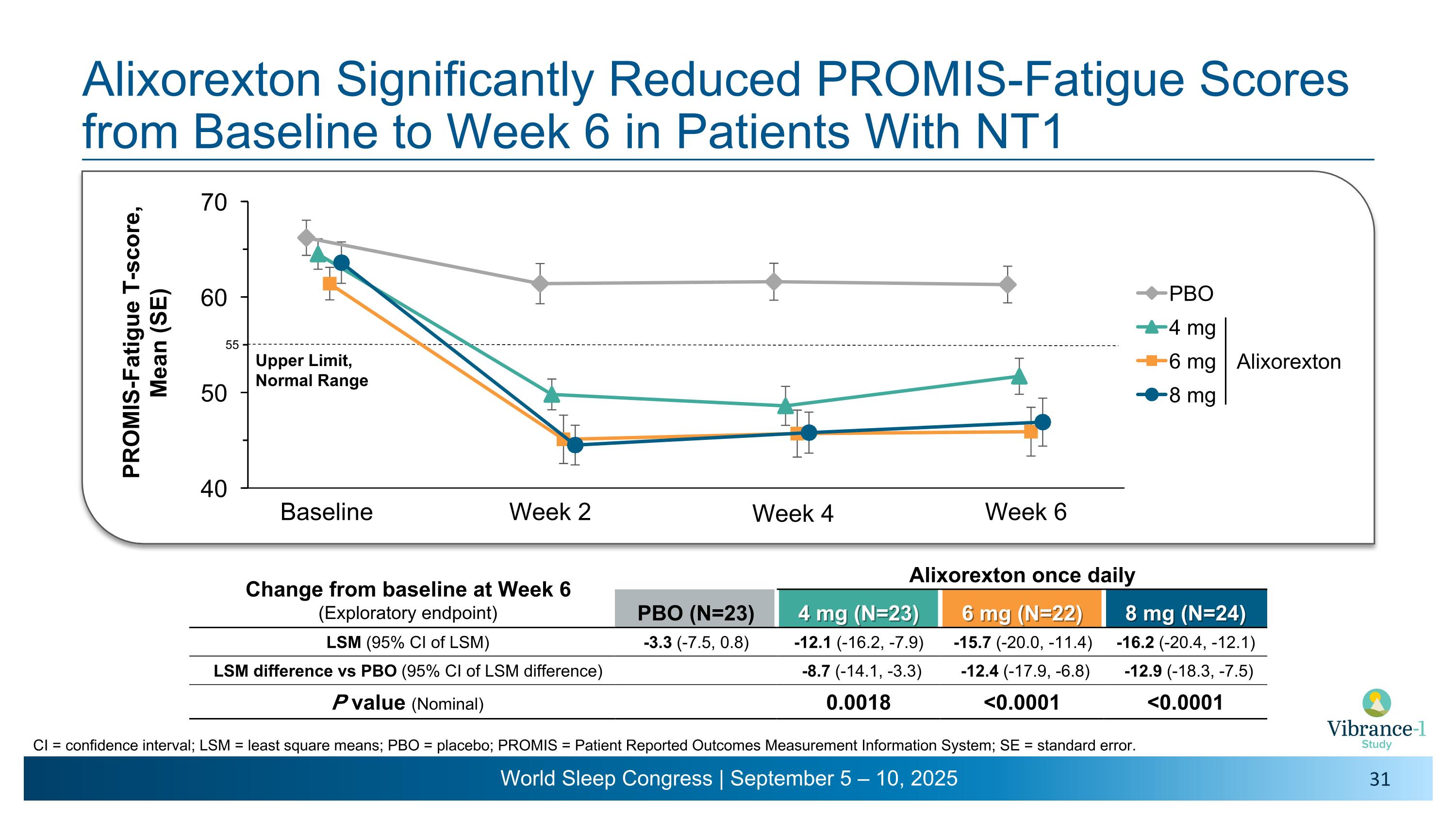
Alixorexton Significantly Reduced PROMIS-Fatigue Scores from Baseline to Week 6 in Patients With NT1 Baseline Week 2 Week 4 Week 6 CI = confidence interval; LSM = least square means; PBO = placebo; PROMIS = Patient Reported Outcomes Measurement Information System; SE = standard error. Upper Limit, Normal Range Change from baseline at Week 6 (Exploratory endpoint) Alixorexton once daily PBO (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) LSM (95% CI of LSM) -3.3 (-7.5, 0.8) -12.1 (-16.2, -7.9) -15.7 (-20.0, -11.4) -16.2 (-20.4, -12.1) LSM difference vs PBO (95% CI of LSM difference) -8.7 (-14.1, -3.3) -12.4 (-17.9, -6.8) -12.9 (-18.3, -7.5) P value (Nominal) 0.0018 <0.0001 <0.0001 55 Alixorexton
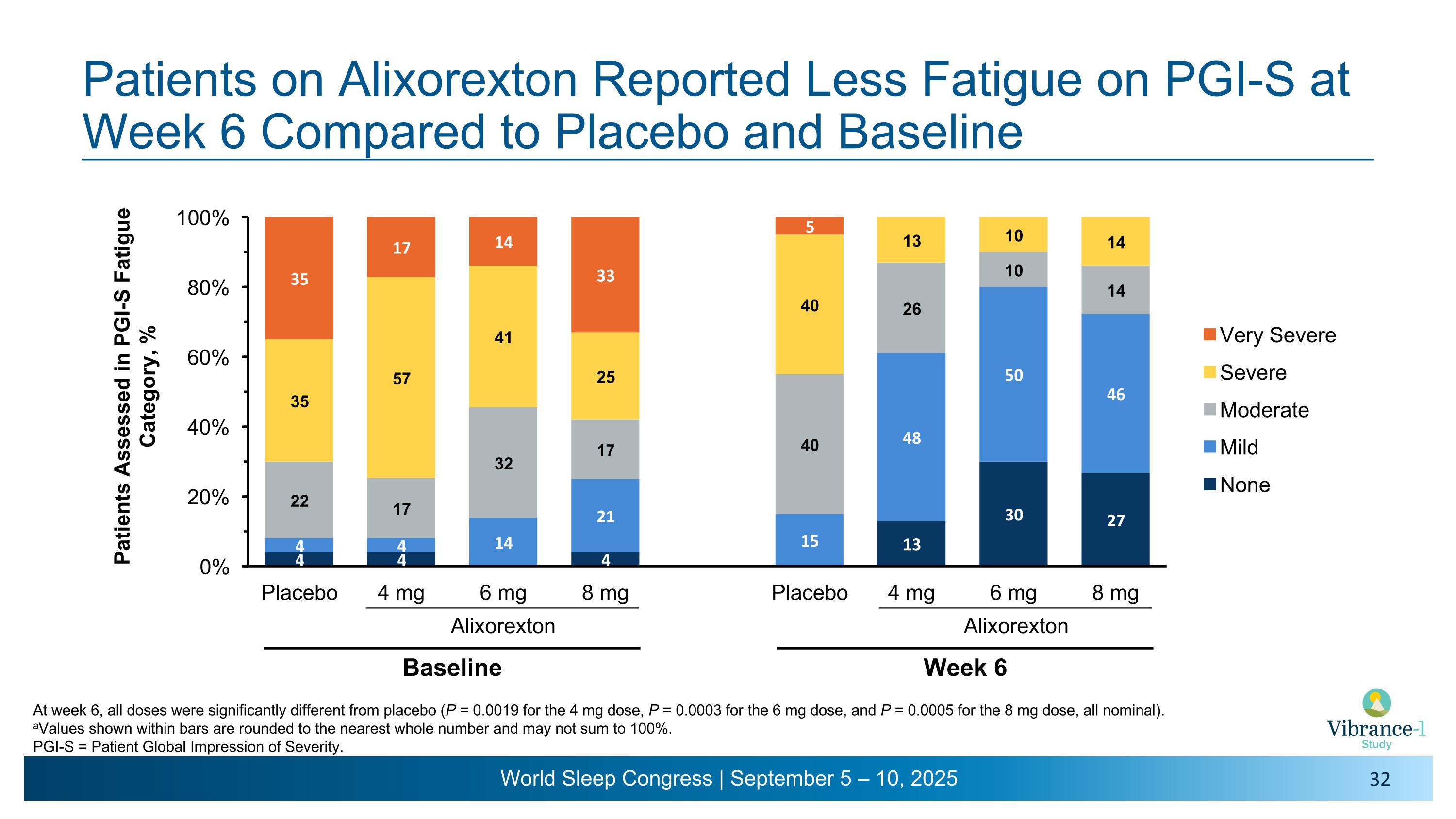
Patients on Alixorexton Reported Less Fatigue on PGI-S at Week 6 Compared to Placebo and Baseline At week 6, all doses were significantly different from placebo (P = 0.0019 for the 4 mg dose, P = 0.0003 for the 6 mg dose, and P = 0.0005 for the 8 mg dose, all nominal). aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. PGI-S = Patient Global Impression of Severity. Alixorexton Baseline Alixorexton Week 6

Alixorexton demonstrated statistically significant, clinically meaningful improvements on established scales: Clinician- and patient-reported severity of narcolepsy symptoms NSS-CT, PGI-S Sleep Disorder, and CGI-S Patient-reported fatigue PROMIS-Fatigue and PGI-S Fatigue Conclusions Alixorexton is the first orexin 2 receptor agonist demonstrating normalized fatigue scores in addition to clinically meaningful improvements �in severity of symptoms in patients with NT1
Alixorexton May Address Many of the Clinical Needs of Patients with NT1, Including Overall Disease Severity and Fatigue BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Acknowledgments The authors would like to thank: The patients Their families and caregivers Vibrance-1 investigators and their staff Funding provided by Alkermes

Giuseppe Plazzi,1 Yves Dauvilliers,2 Ronald R. Grunstein,3 Emmanuel Mignot,4 Gert Jan Lammers,5 David T. Plante,6 Erik Buntinx,7 Rafael del Río Villegas,8 Hailu Chen,9 Craig Hopkinson,9 Bhaskar Rege,9 Marcus Yountz,9 and Michael J. Doane9 Improvement in Patient-reported Cognitive Functioning in Patients with Narcolepsy Type 1 Treated With the Orexin 2 Receptor Agonist Alixorexton (ALKS 2680) 1IRCCS Istituto delle Scienze Neurologiche di Bologna, Bologna, Italy; 2University of Montpellier, INSERM Institute for Neurosciences of Montpellier, Montpellier, France; 3Woolcock Institute of Medical Research, Macquarie University, Sydney, Australia; 4Stanford Center for Sleep Sciences and Medicine, Stanford University School of Medicine, Stanford, CA, USA; 5Leiden University Medical Center, Leiden, the Netherlands and Stichting Epilepsie Instellingen Nederland, Sleep-Wake Centre, Heemstede, the Netherlands; 6University of Wisconsin-Madison, School of Medicine and Public Health, Madison, WI, USA; 7ANIMA Research, Alken, Belgium; 8Universidad CEU San Pablo, CEU Universities, Vithas Madrid Hospitals Madrid, Spain; 9Alkermes, Inc., Waltham, MA, USA

Ineligible companies are those whose primary business is producing, marketing, selling, re-selling, or distributing health care products used by, or on patients. ◻︎ No, I HAVE NOT had a financial relationship with an ineligible company in the past 24 months. ◻︎ Yes, I HAVE had a financial relationship with an ineligible company in the past 24 months. Relationship type Name of company Institutional funding Alkermes (R. Grunstein, Y. Dauvilliers); Lilly (R. Grunstein); Takeda (R. Grunstein); Vanda (R. Grunstein) Research funding Avadel (E. Mignot); Alkermes (E. Mignot); Bioprojet (G. Plazzi); Centessa Pharmaceuticals (G. Plazzi); Eisai (E. Mignot); Idorsia (G. Plazzi); Jazz Pharmaceuticals (G. Plazzi, E. Mignot); Orexia Therapeutics (G. Plazzi); Takeda (G. Plazzi, E. Mignot); Vanda (E. Mignot) Employment Alkermes (B. Rege, C. Hopkinson, H. Chen, M. Doane, M. Yountz) Speaker fees Eisai (R. Grunstein); SomnoMed (R. Grunstein) Advisory Board Aditum Bio LLC (D. Plante); Alkermes (D. Plante): Apnimed (R. Grunstein); Avadel (Y. Dauvilliers); Bioprojet (Y. Dauvilliers); Centessa (D. Plante, Y. Dauvilliers); Harmony Biosciences (D. Plante, Y. Dauvilliers); Idorsia (Bioprojet); Jazz Pharmaceuticals (D. Plante, Y. Dauvilliers); Lilly (R. Grunstein); Takeda (D. Plante, E. Mignot, Y. Dauvilliers); TEVA (D. Plante).
Vibrance-1 Examined the Effects of Alixorexton on Cognitive Impairment in Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Patients Describe Negative Impacts Across: Memory Problem Solving Attention Processing Speed Word Finding Expressing Thoughts Beyond sleepiness and cataplexy, people with narcolepsy frequently report experiencing cognitive impairment and overall “brain fog”1-3 There are negative impacts of cognitive impairment across individuals’ lives, including to school/work, relationships, risk of accidents, and quality of life2-5 Cognitive impairment may persist even when excessive daytime sleepiness is adequately controlled4,6 Cognitive Impairment is Among the Most Common and Disruptive Symptoms Reported by People With Narcolepsy 1. Cano C, et al. Sleep. 2024;47(9):zsae150. 2. Doane M, et al. Poster presented at US Psych 2023. 3. Maski K, et al. J Clin Sleep Med. 2017;13(3):419-425. 4. Harel B et al. Sleep Advances. 2024; 26;5(1):zpae043. 5. Blackwell J, et al. Sleep Medicine Reviews. 2017;34:82-93. 6. Winter Y, et al. Poster presented at APSS 2024.

OX2R Agonism May Enhance Cognition by Activating Receptors Expressed Throughout Cognitive Circuitry 1. Marcus JN, et al. J Comp Neurol. 2001;435(1):6-25. 2. Sakurai T. Nat Rev Neurosci. 2007;8(3):171-181. 3. Alexandre C, et al. Curr Opin Neurobiol. 2013;23(5):752-759. 4. Sarter M, et al. Brain Res Rev. 2006;51(2):145-160. 5. Katzman MA, et al. Brain Sci. 2022;12(2):150. AM = amygdala; BF = basal forebrain; DR = dorsal raphe; NAc = nucleus accumbens; OX2R = orexin 2 receptor; SN = substantia nigra; TH = thalamus; TMN = tuberomammillary nucleus; VTA = ventral tegmental area. Orexin pathway �Originates in hypothalamus and engages distributed neuronal circuitry Promotes adaptive behavioral responses
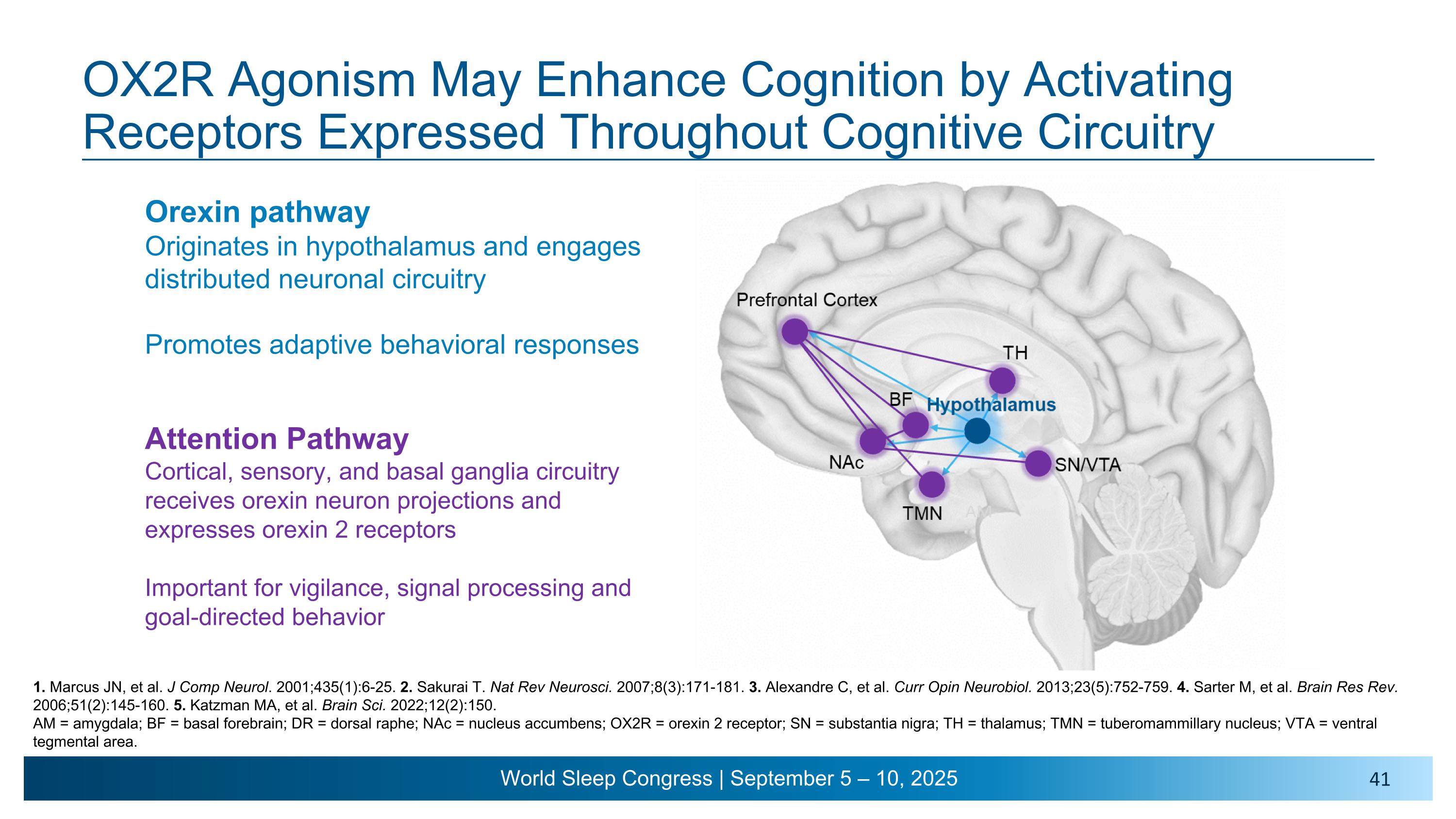
OX2R Agonism May Enhance Cognition by Activating Receptors Expressed Throughout Cognitive Circuitry Orexin pathway �Originates in hypothalamus and engages distributed neuronal circuitry Promotes adaptive behavioral responses Attention Pathway Cortical, sensory, and basal ganglia circuitry receives orexin neuron projections and expresses orexin 2 receptors Important for vigilance, signal processing and goal-directed behavior 1. Marcus JN, et al. J Comp Neurol. 2001;435(1):6-25. 2. Sakurai T. Nat Rev Neurosci. 2007;8(3):171-181. 3. Alexandre C, et al. Curr Opin Neurobiol. 2013;23(5):752-759. 4. Sarter M, et al. Brain Res Rev. 2006;51(2):145-160. 5. Katzman MA, et al. Brain Sci. 2022;12(2):150. AM = amygdala; BF = basal forebrain; DR = dorsal raphe; NAc = nucleus accumbens; OX2R = orexin 2 receptor; SN = substantia nigra; TH = thalamus; TMN = tuberomammillary nucleus; VTA = ventral tegmental area.
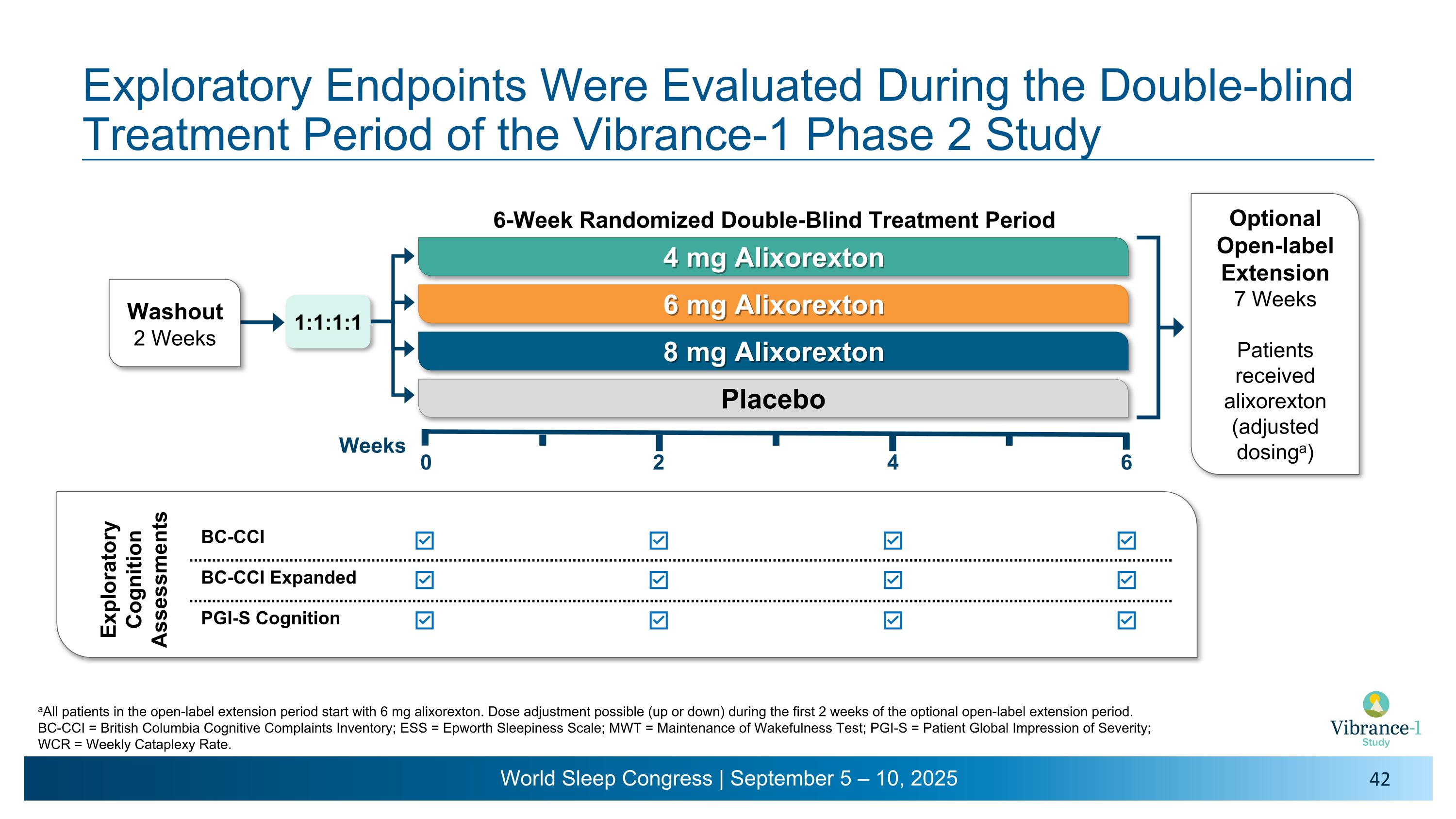
0 Weeks Optional Open-label Extension 7 Weeks Patients received alixorexton �(adjusted dosinga) Exploratory Endpoints Were Evaluated During the Double-blind Treatment Period of the Vibrance-1 Phase 2 Study 4 mg Alixorexton 6 mg Alixorexton 8 mg Alixorexton Placebo 6-Week Randomized Double-Blind Treatment Period 1:1:1:1 2 4 6 BC-CCI BC-CCI Expanded PGI-S Cognition Washout 2 Weeks aAll patients in the open-label extension period start with 6 mg alixorexton. Dose adjustment possible (up or down) during the first 2 weeks of the optional open-label extension period. BC-CCI = British Columbia Cognitive Complaints Inventory; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; PGI-S = Patient Global Impression of Severity; �WCR = Weekly Cataplexy Rate. Exploratory Cognition Assessments

Cognitive Impairment BC-CCI Assesses the Severity and Impact of Cognitive Impairment From the Patient’s Perspective BC-CCI = British Columbia Cognitive Complaints Inventory. Items include: Forgetfulness/memory problems Poor concentration Trouble expressing thoughts Trouble finding the right word Slow thinking speed Trouble figuring things out or solving problems Perceived Severity Rating Categories Severe 15 – 18 Moderate 9 – 14 Mild 5 – 8 None or Minimal 0 – 4
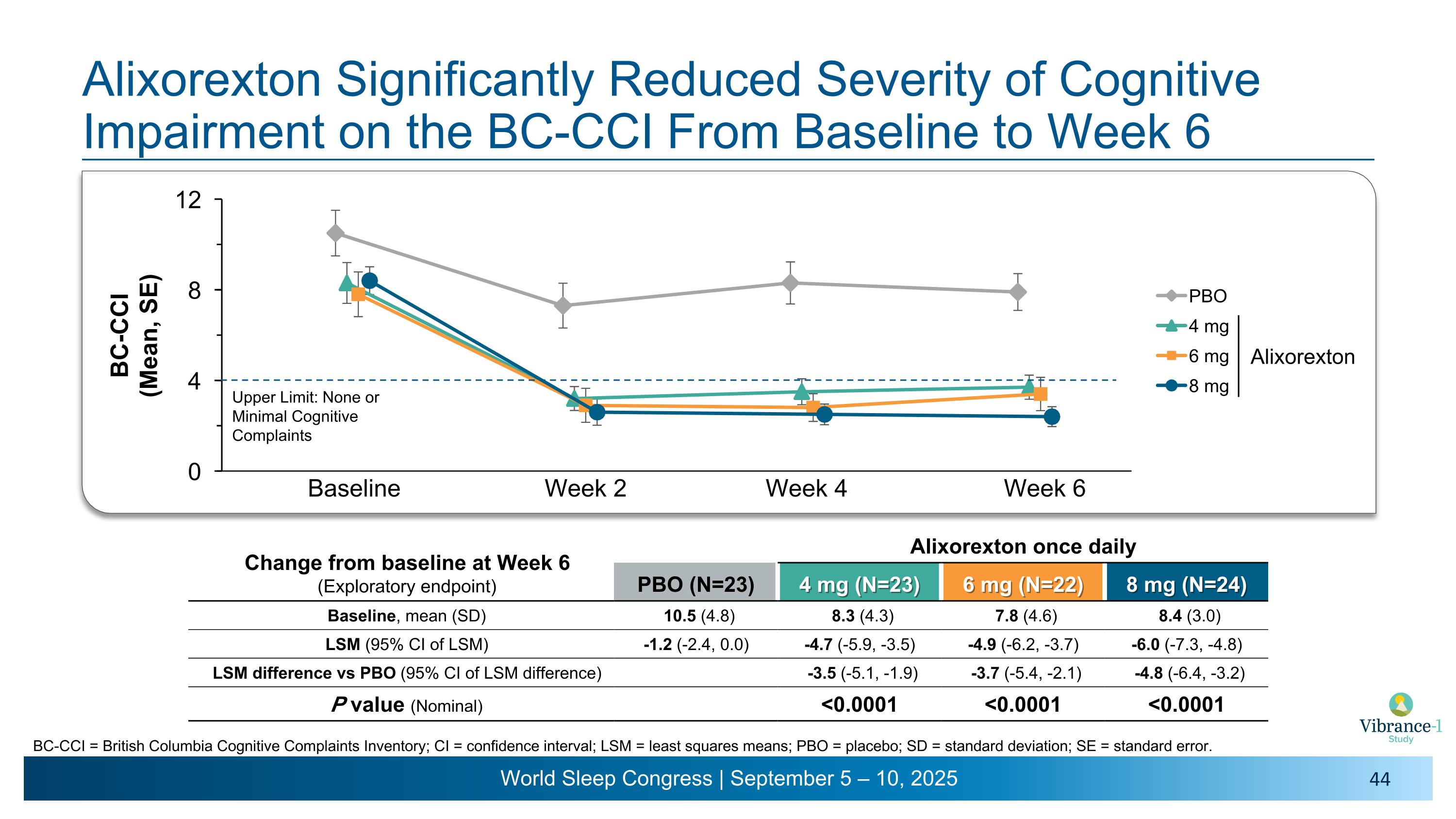
Baseline Week 2 Week 4 Week 6 Alixorexton Significantly Reduced Severity of Cognitive Impairment on the BC-CCI From Baseline to Week 6 BC-CCI = British Columbia Cognitive Complaints Inventory; CI = confidence interval; LSM = least squares means; PBO = placebo; SD = standard deviation; SE = standard error. Upper Limit: None or Minimal Cognitive Complaints Change from baseline at Week 6 (Exploratory endpoint) Alixorexton once daily PBO (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Baseline, mean (SD) 10.5 (4.8) 8.3 (4.3) 7.8 (4.6) 8.4 (3.0) LSM (95% CI of LSM) -1.2 (-2.4, 0.0) -4.7 (-5.9, -3.5) -4.9 (-6.2, -3.7) -6.0 (-7.3, -4.8) LSM difference vs PBO (95% CI of LSM difference) -3.5 (-5.1, -1.9) -3.7 (-5.4, -2.1) -4.8 (-6.4, -3.2) P value (Nominal) <0.0001 <0.0001 <0.0001 Alixorexton
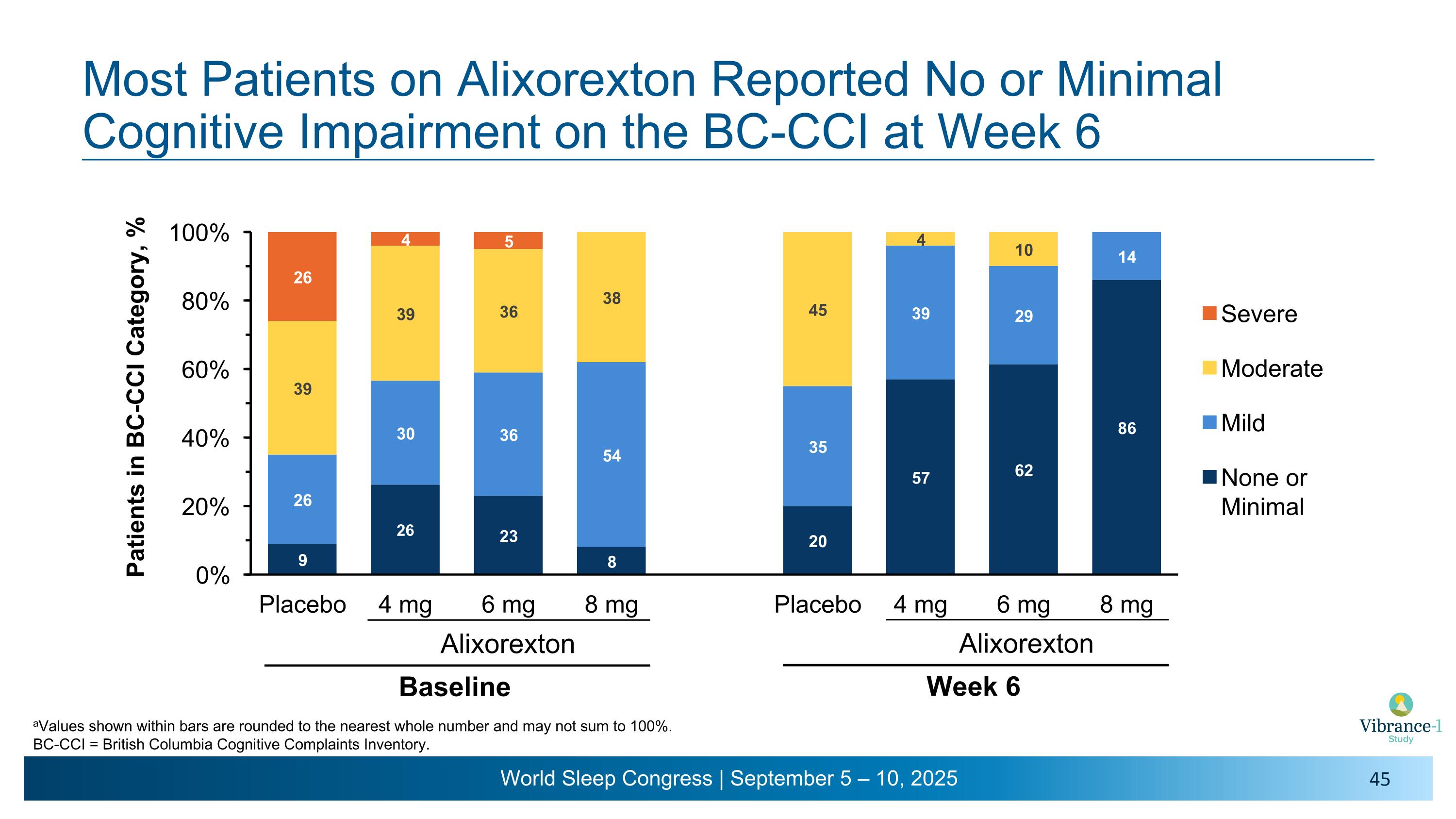
Most Patients on Alixorexton Reported No or Minimal Cognitive Impairment on the BC-CCI at Week 6 Alixorexton Baseline Alixorexton Week 6 aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. BC-CCI = British Columbia Cognitive Complaints Inventory.
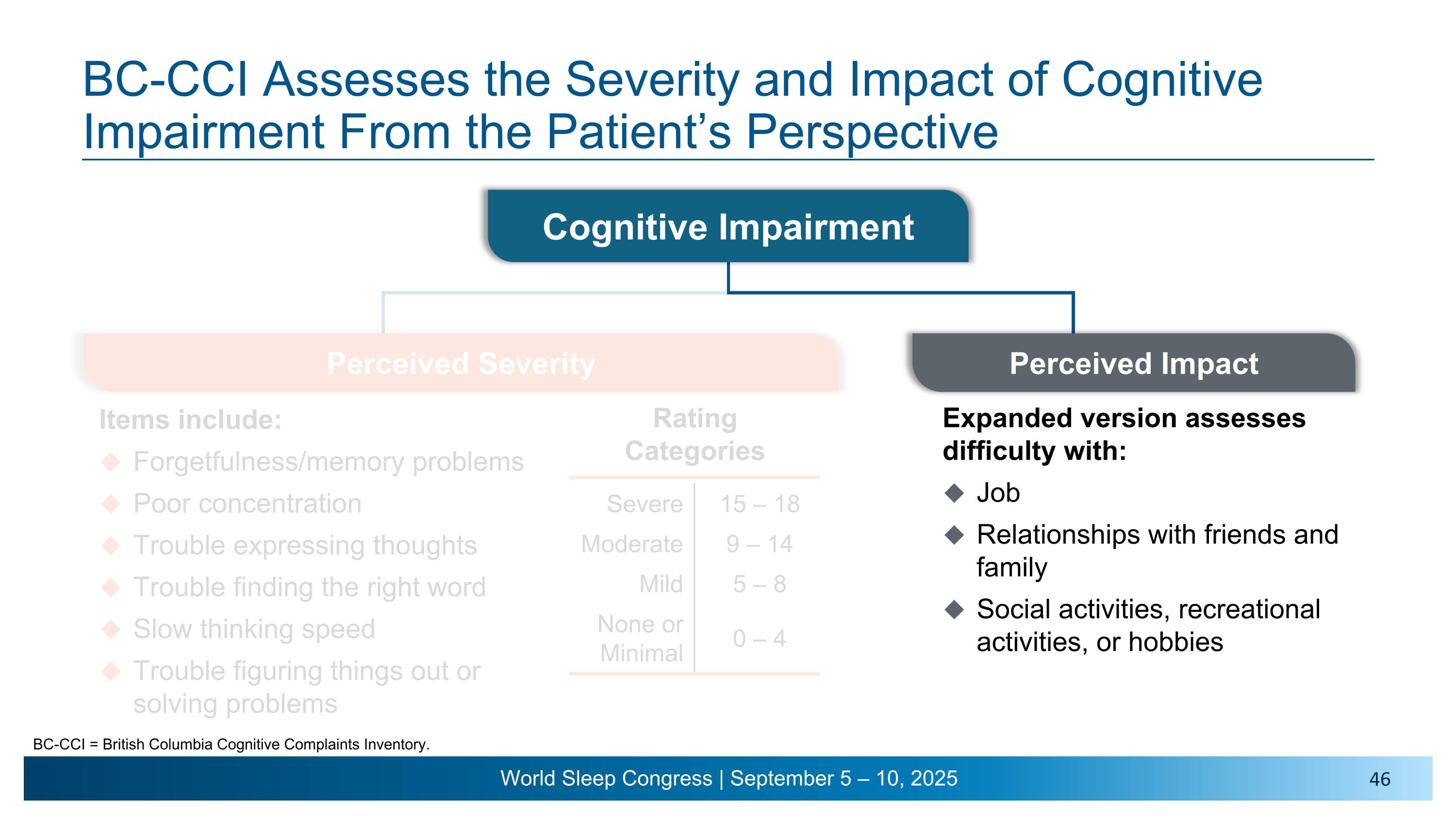
BC-CCI Assesses the Severity and Impact of Cognitive Impairment From the Patient’s Perspective Items include: Forgetfulness/memory problems Poor concentration Trouble expressing thoughts Trouble finding the right word Slow thinking speed Trouble figuring things out or solving problems Perceived Severity Perceived Impact Expanded version assesses difficulty with: Job Relationships with friends and family Social activities, recreational activities, or hobbies Rating Categories Severe 15 – 18 Moderate 9 – 14 Mild 5 – 8 None or Minimal 0 – 4 Cognitive Impairment BC-CCI = British Columbia Cognitive Complaints Inventory.
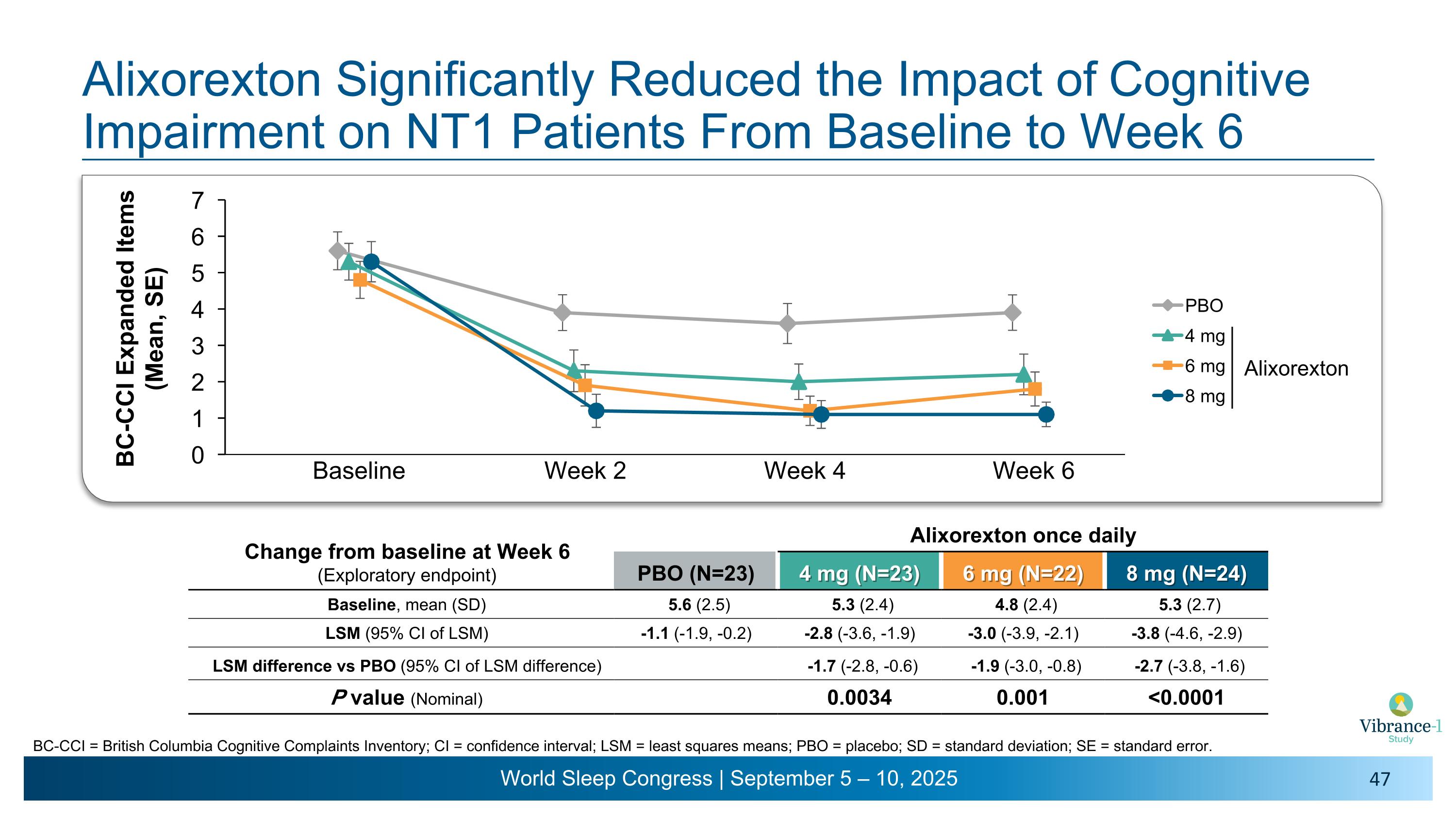
Alixorexton Significantly Reduced the Impact of Cognitive Impairment on NT1 Patients From Baseline to Week 6 Baseline Week 2 Week 4 Week 6 Change from baseline at Week 6�(Exploratory endpoint) Alixorexton once daily PBO (N=23) 4 mg (N=23) 6 mg (N=22) 8 mg (N=24) Baseline, mean (SD) 5.6 (2.5) 5.3 (2.4) 4.8 (2.4) 5.3 (2.7) LSM (95% CI of LSM) -1.1 (-1.9, -0.2) -2.8 (-3.6, -1.9) -3.0 (-3.9, -2.1) -3.8 (-4.6, -2.9) LSM difference vs PBO (95% CI of LSM difference) -1.7 (-2.8, -0.6) -1.9 (-3.0, -0.8) -2.7 (-3.8, -1.6) P value (Nominal) 0.0034 0.001 <0.0001 Alixorexton BC-CCI = British Columbia Cognitive Complaints Inventory; CI = confidence interval; LSM = least squares means; PBO = placebo; SD = standard deviation; SE = standard error.
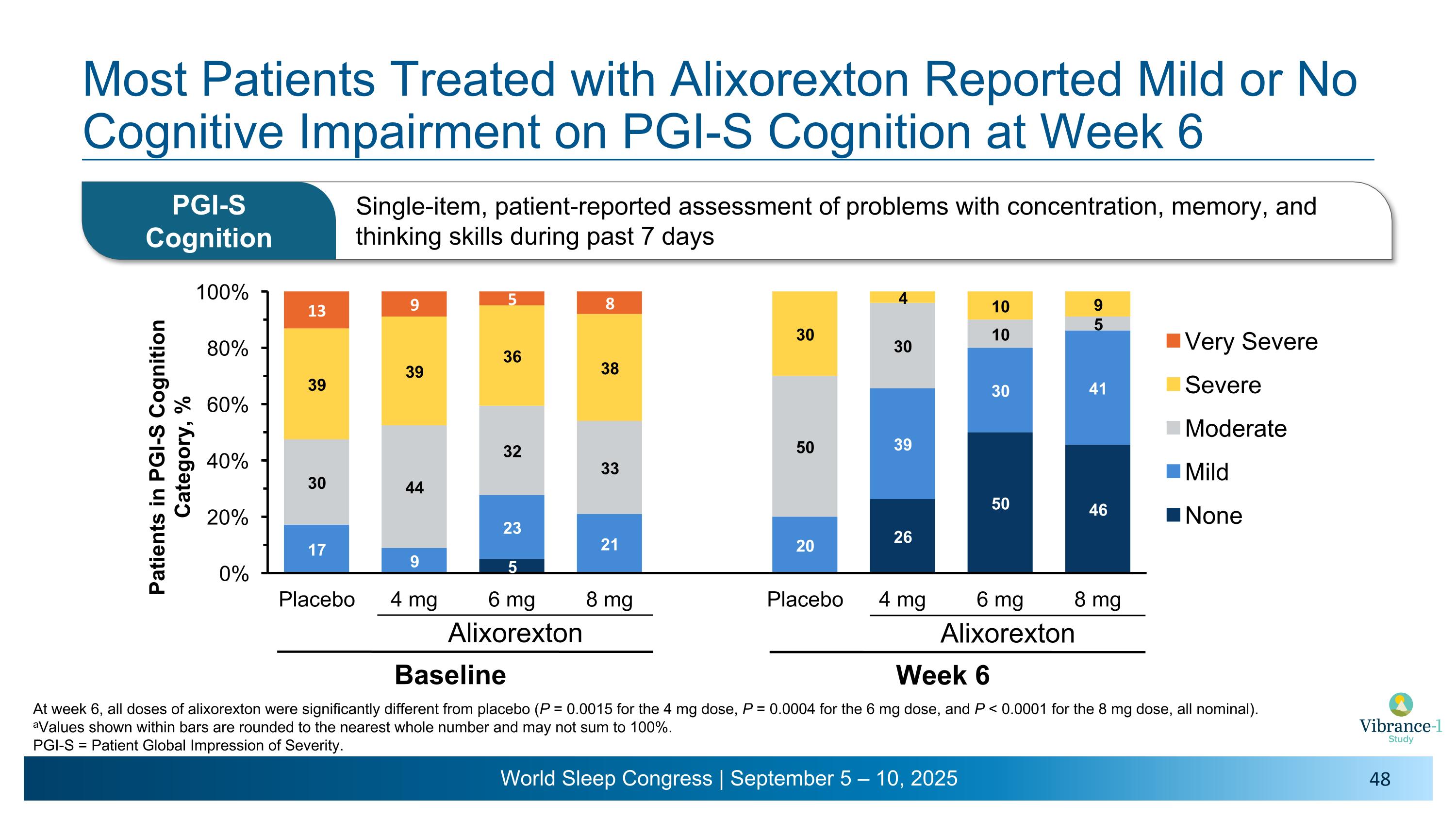
PGI-S Cognition Most Patients Treated with Alixorexton Reported Mild or No Cognitive Impairment on PGI-S Cognition at Week 6 At week 6, all doses of alixorexton were significantly different from placebo (P = 0.0015 for the 4 mg dose, P = 0.0004 for the 6 mg dose, and P < 0.0001 for the 8 mg dose, all nominal). aValues shown within bars are rounded to the nearest whole number and may not sum to 100%. PGI-S = Patient Global Impression of Severity. Single-item, patient-reported assessment of problems with concentration, memory, and thinking skills during past 7 days Alixorexton Baseline Alixorexton Week 6

Conclusions Alixorexton demonstrated statistically significant and clinically meaningful improvements from baseline on established measures evaluating the severity and impact of cognitive impairment in patients with NT1 BC-CCI: improvements observed on overall patient-reported outcome measure encompassing a range of cognitive domains Severity: memory, attention, word finding, problem solving, processing speed, and expressing thoughts Impact: work, relationships and daily activities (expanded items) PGI-S: findings of patient-reported severity consistent with BC-CCI Alixorexton is the first OX2R agonist to demonstrate normalization of �cognitive functioning on patient-reported scales in the NT1 population

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Statistically significant, clinically meaningful improvements compared to placebo on the MWT and ESS, achieving a normative wakefulness profile BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Statistically significant, clinically meaningful improvements compared to placebo on the MWT and ESS, achieving a normative wakefulness profile Clinically meaningful improvement in WCR at all doses compared to placebo, with statistical significance achieved at the 6 mg dose BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Statistically significant, clinically meaningful improvements compared to placebo on the MWT and ESS, achieving a normative wakefulness profile Clinically meaningful improvement in WCR at all doses compared to placebo, with statistical significance achieved at the 6 mg dose Generally well tolerated, with most TEAEs mild to moderate in severity and no serious TEAEs reported BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Statistically significant, clinically meaningful improvements compared to placebo on the MWT and ESS, achieving a normative wakefulness profile Clinically meaningful improvement in WCR at all doses compared to placebo, with statistical significance achieved at the 6 mg dose Generally well tolerated, with most TEAEs mild to moderate in severity and no serious TEAEs reported Statistically significant, clinically meaningful improvements compared to placebo on patient-and clinician-reported narcolepsy symptom severity BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Statistically significant, clinically meaningful improvements compared to placebo on the MWT and ESS, achieving a normative wakefulness profile Clinically meaningful improvement in WCR at all doses compared to placebo, with statistical significance achieved at the 6 mg dose Generally well tolerated, with most TEAEs mild to moderate in severity and no serious TEAEs reported Statistically significant, clinically meaningful improvements compared to placebo on patient-and clinician-reported narcolepsy symptom severity Statistically significant, clinically meaningful improvements on two established patient-reported fatigue scales compared to placebo BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.
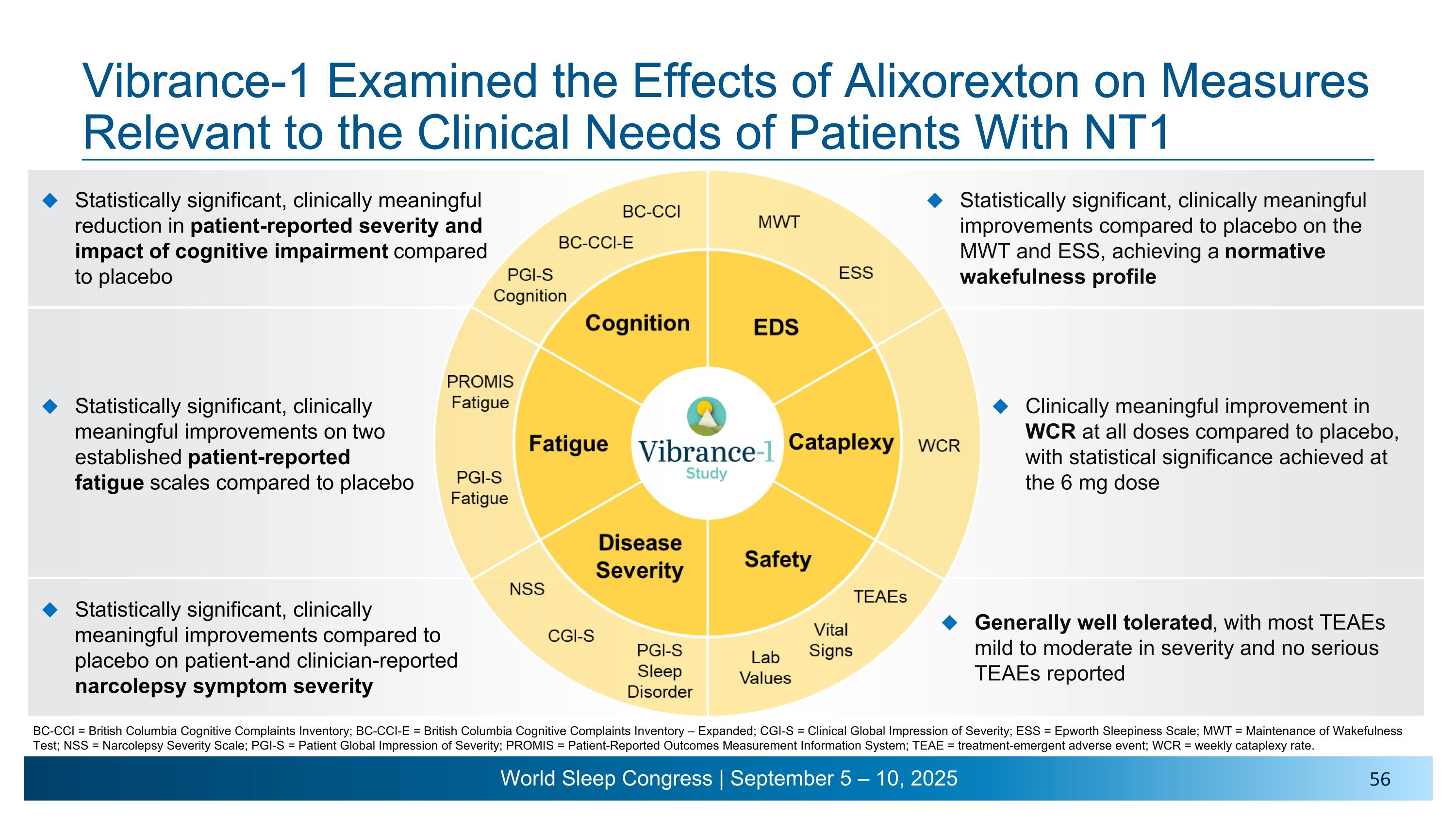
Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Vibrance-1 Examined the Effects of Alixorexton on Measures Relevant to the Clinical Needs of Patients With NT1 Statistically significant, clinically meaningful reduction in patient-reported severity and impact of cognitive impairment compared to placebo Statistically significant, clinically meaningful improvements compared to placebo on the MWT and ESS, achieving a normative wakefulness profile Clinically meaningful improvement in WCR at all doses compared to placebo, with statistical significance achieved at the 6 mg dose Generally well tolerated, with most TEAEs mild to moderate in severity and no serious TEAEs reported Statistically significant, clinically meaningful improvements compared to placebo on patient-and clinician-reported narcolepsy symptom severity Statistically significant, clinically meaningful improvements on two established patient-reported fatigue scales compared to placebo BC-CCI = British Columbia Cognitive Complaints Inventory; BC-CCI-E = British Columbia Cognitive Complaints Inventory – Expanded; CGI-S = Clinical Global Impression of Severity; ESS = Epworth Sleepiness Scale; MWT = Maintenance of Wakefulness Test; NSS = Narcolepsy Severity Scale; PGI-S = Patient Global Impression of Severity; PROMIS = Patient-Reported Outcomes Measurement Information System; TEAE = treatment-emergent adverse event; WCR = weekly cataplexy rate.

Once-daily Alixorexton Is Being Evaluated in NT1, NT2, and IH 1. Alkermes, Inc. NCT06358950 (Vibrance-1). https://clinicaltrials.gov/study/NCT06358950. 2. Alkermes, Inc. NCT06555783 (Vibrance-2). https://www.clinicaltrials.gov/study/NCT06555783. 3. Alkermes, Inc. NCT06843590 (Vibrance-3). https://clinicaltrials.gov/study/NCT06843590. NT1 = narcolepsy type 1; NT2 = narcolepsy type 2; IH = idiopathic hypersomnia. NT1 NT2 IH Complete Enrollment complete Enrollment ongoing Results from Vibrance-1 will inform dose selection for the �global phase 3 development program in patients with NT1

Acknowledgments The authors would like to thank: The patients Their families and caregivers Vibrance-1 investigators and their staff Funding provided by Alkermes




























































































